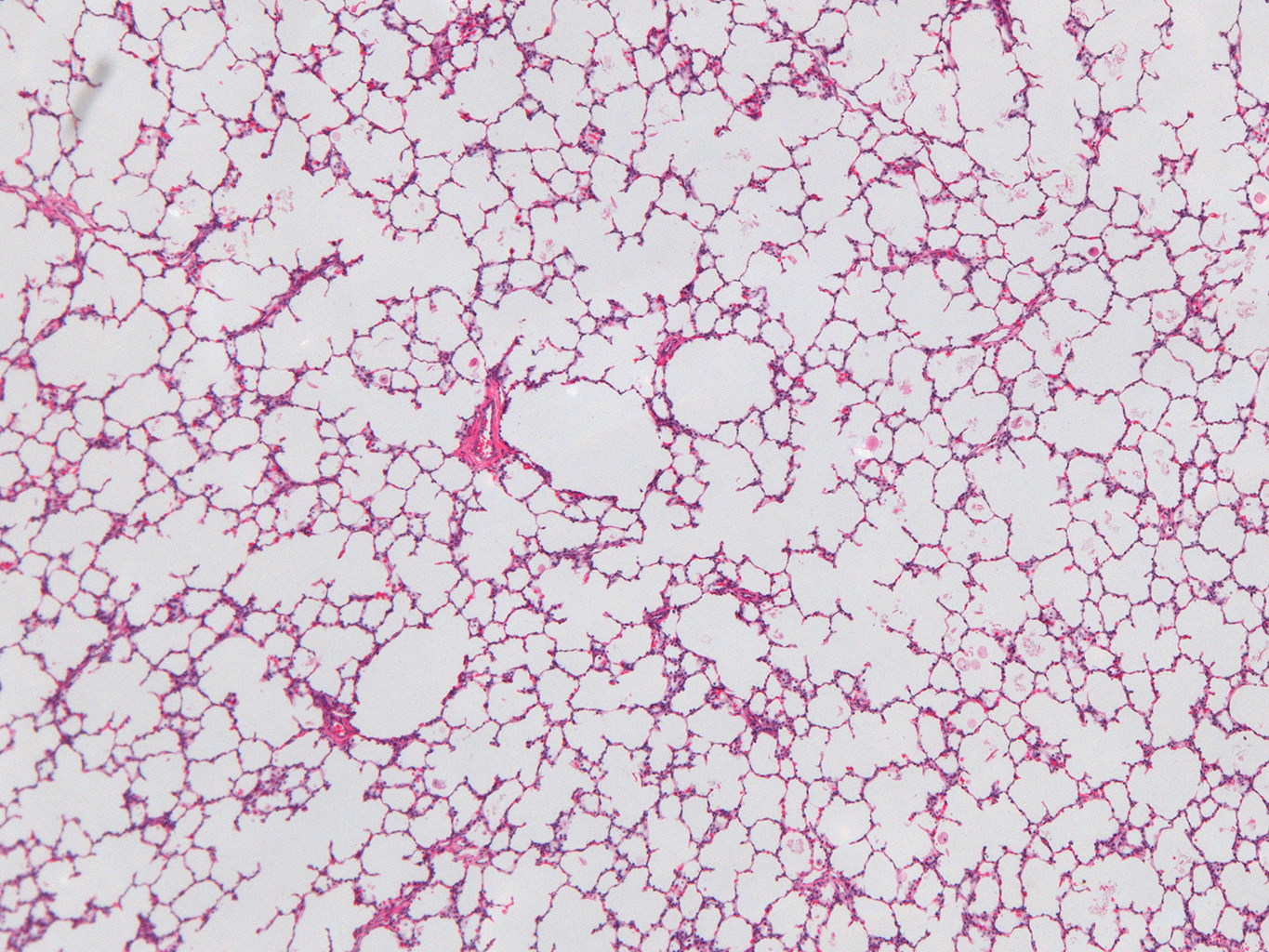
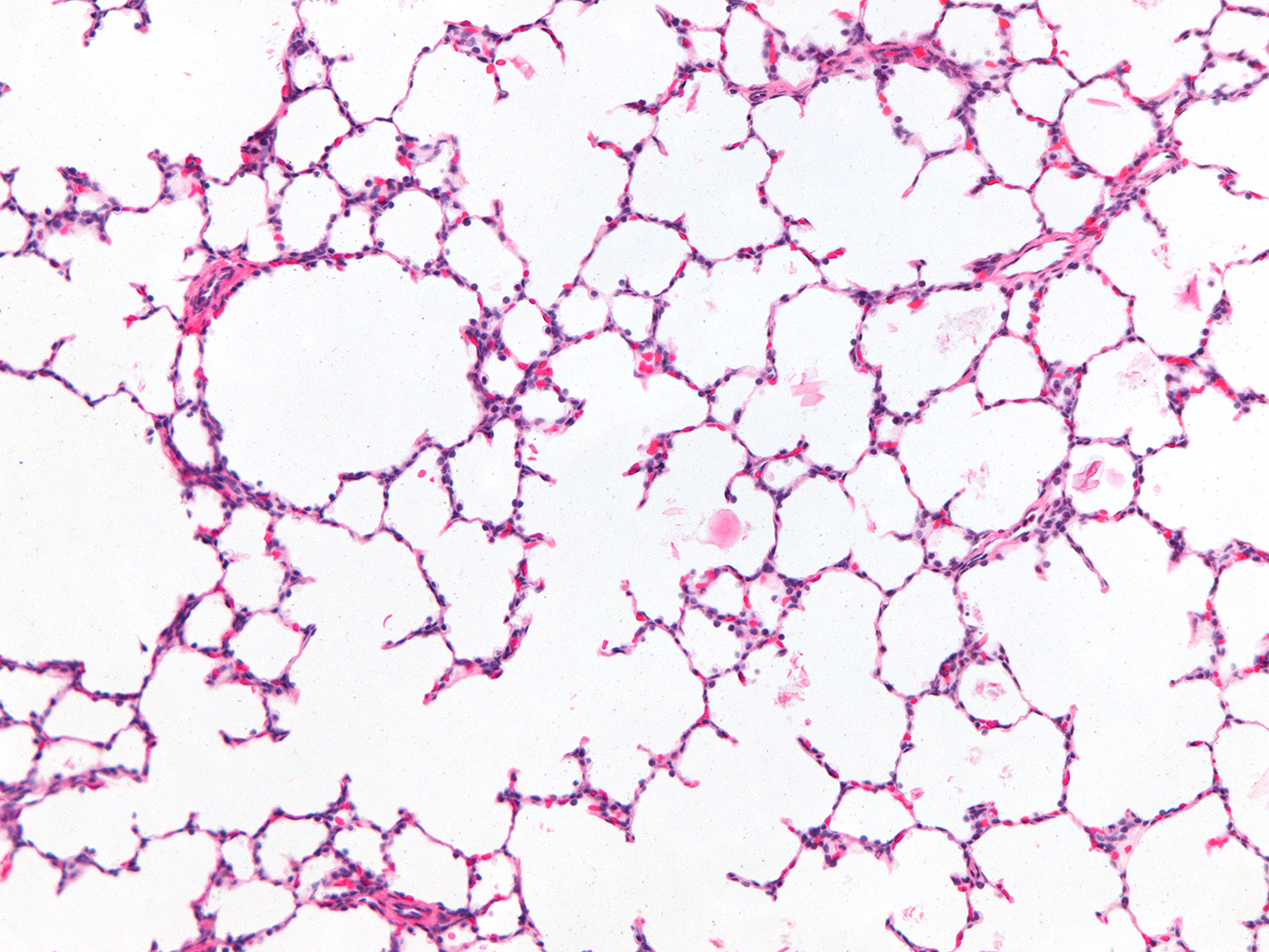
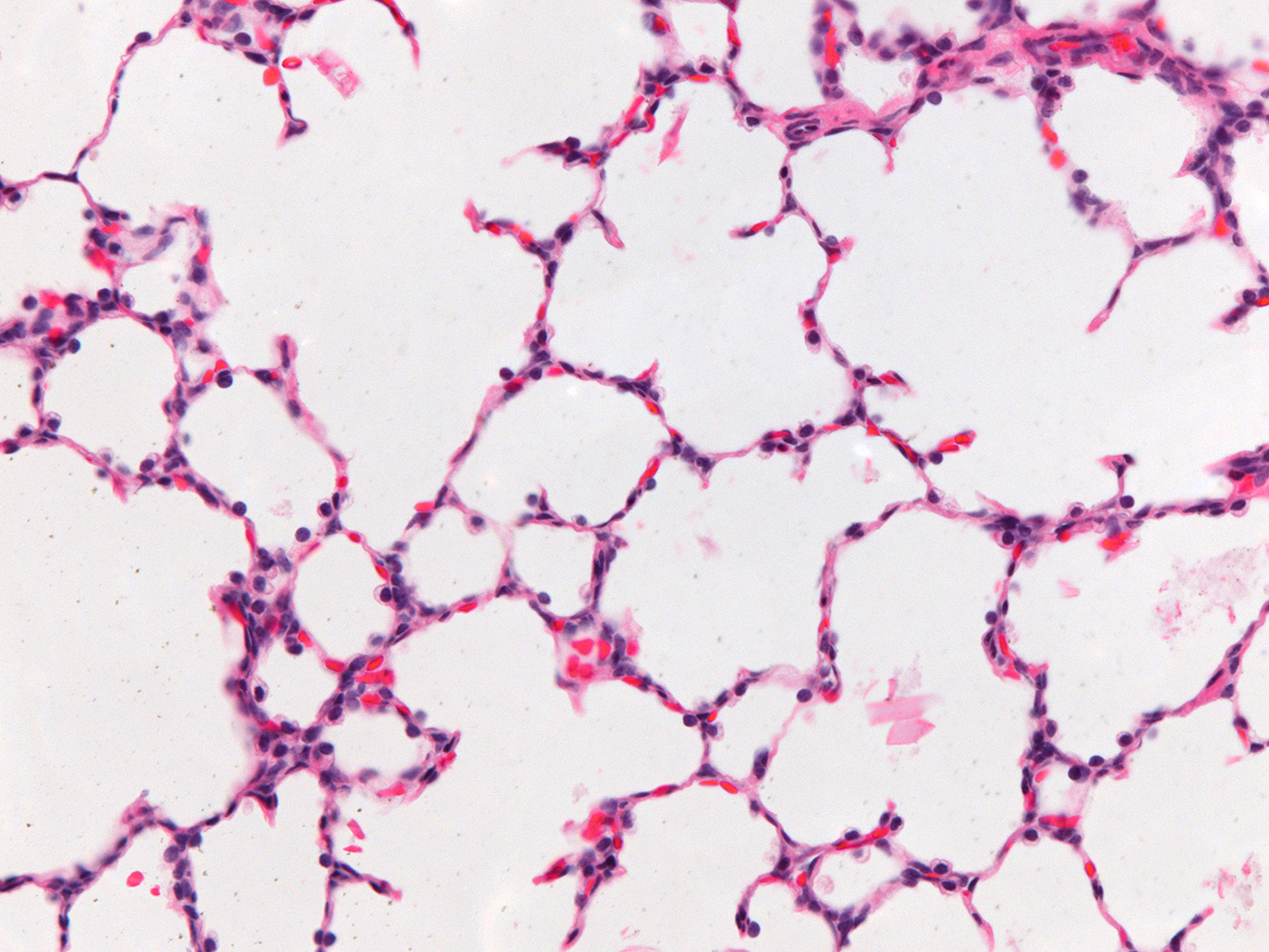
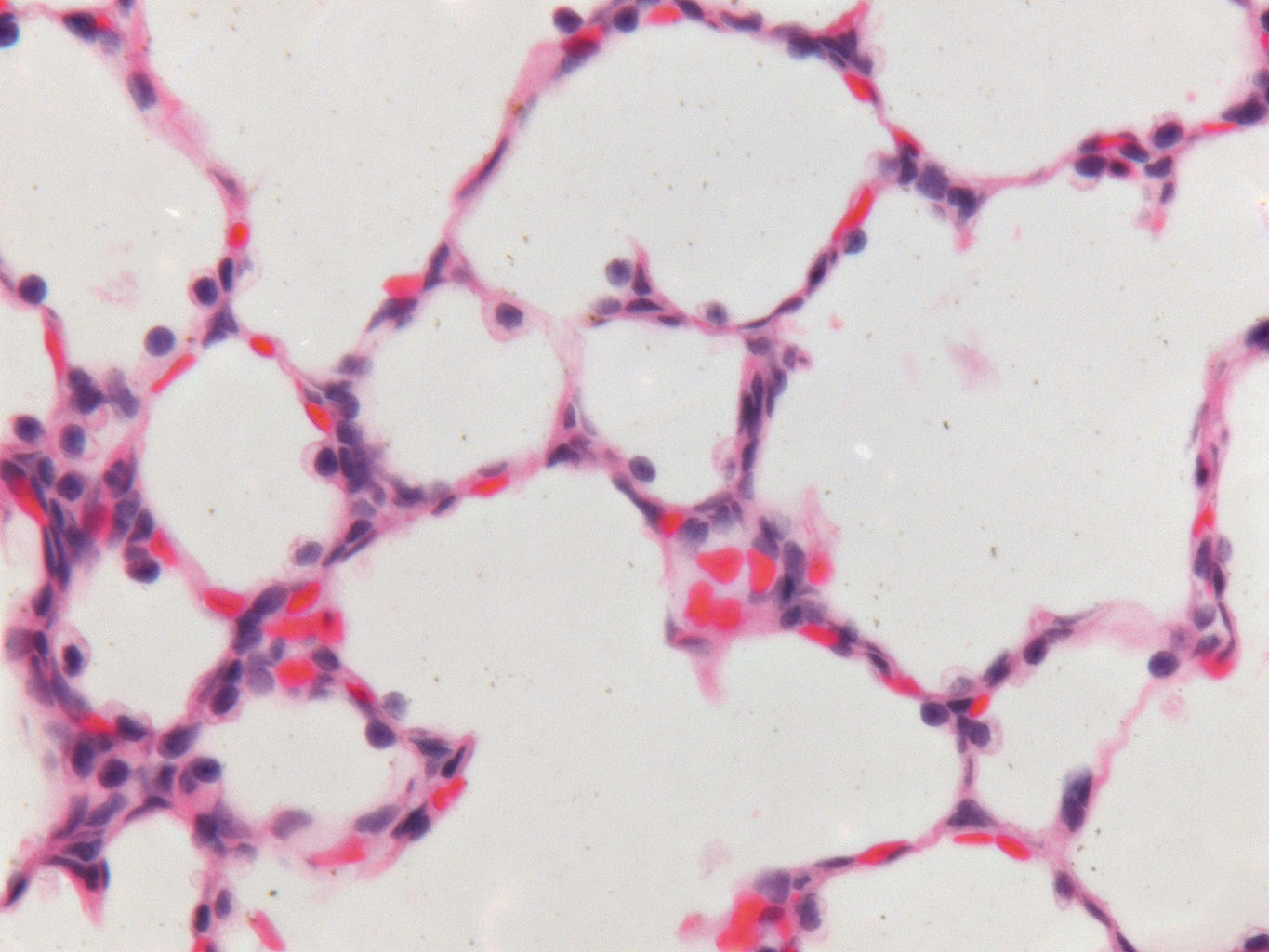
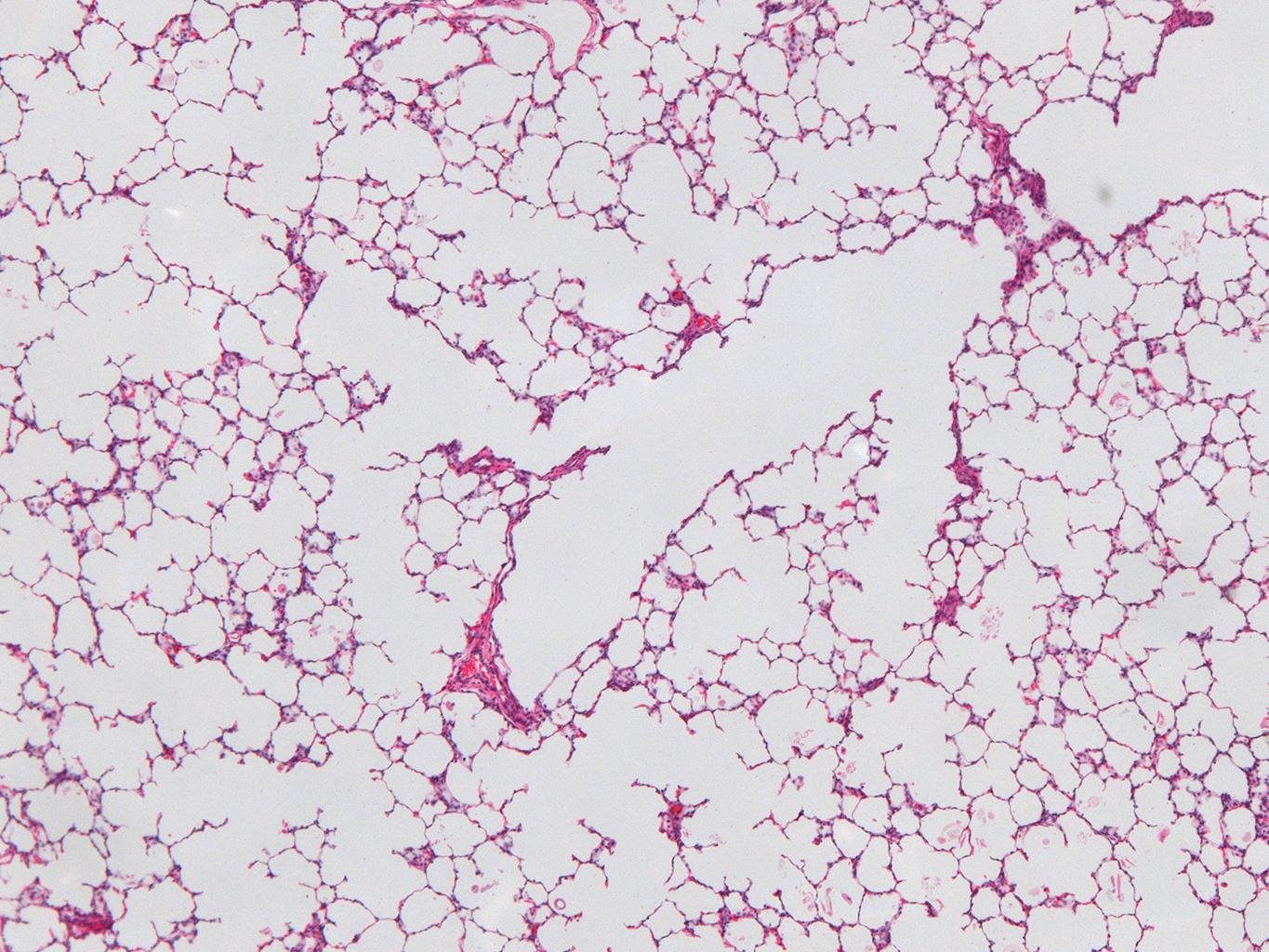
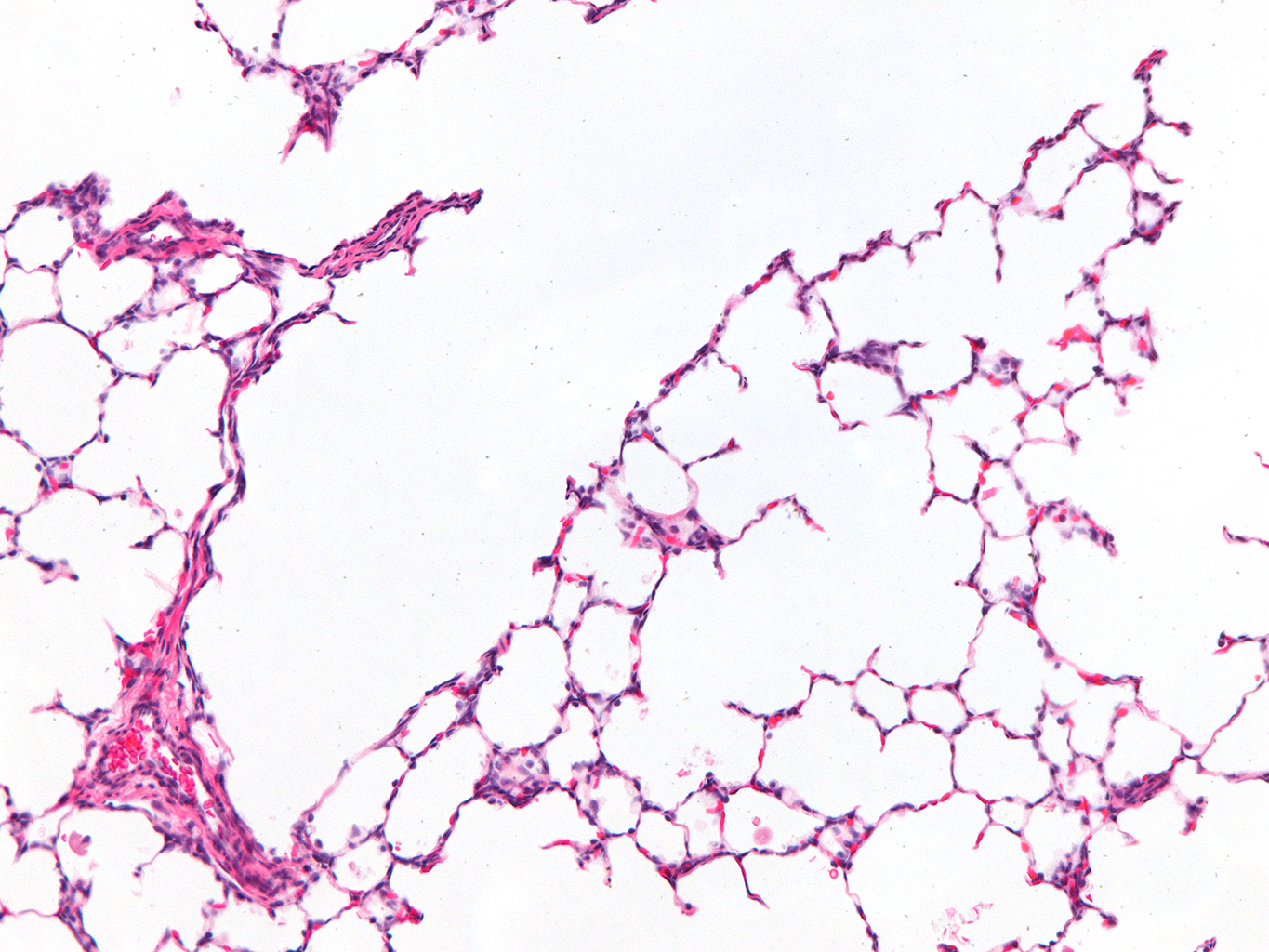
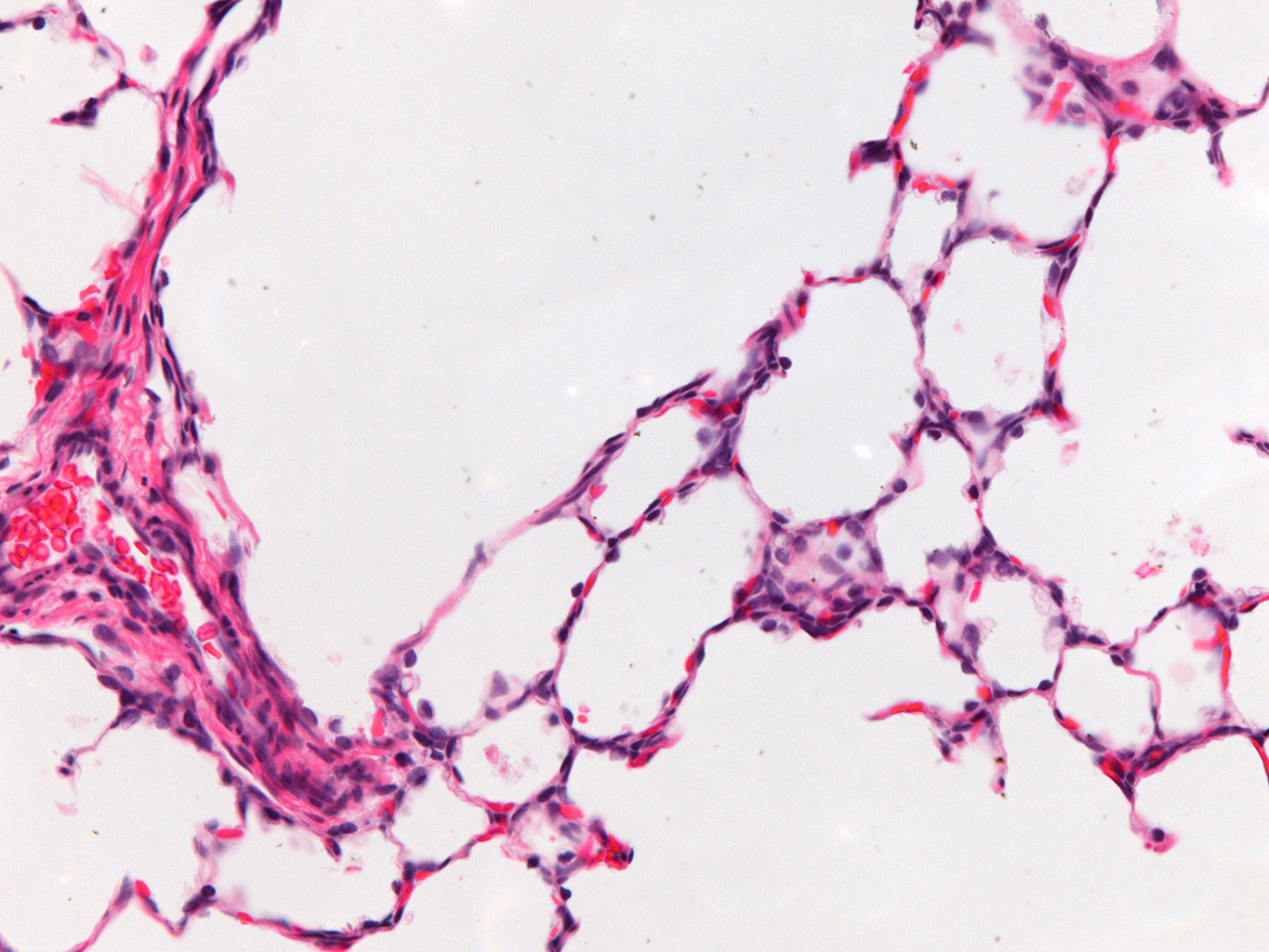
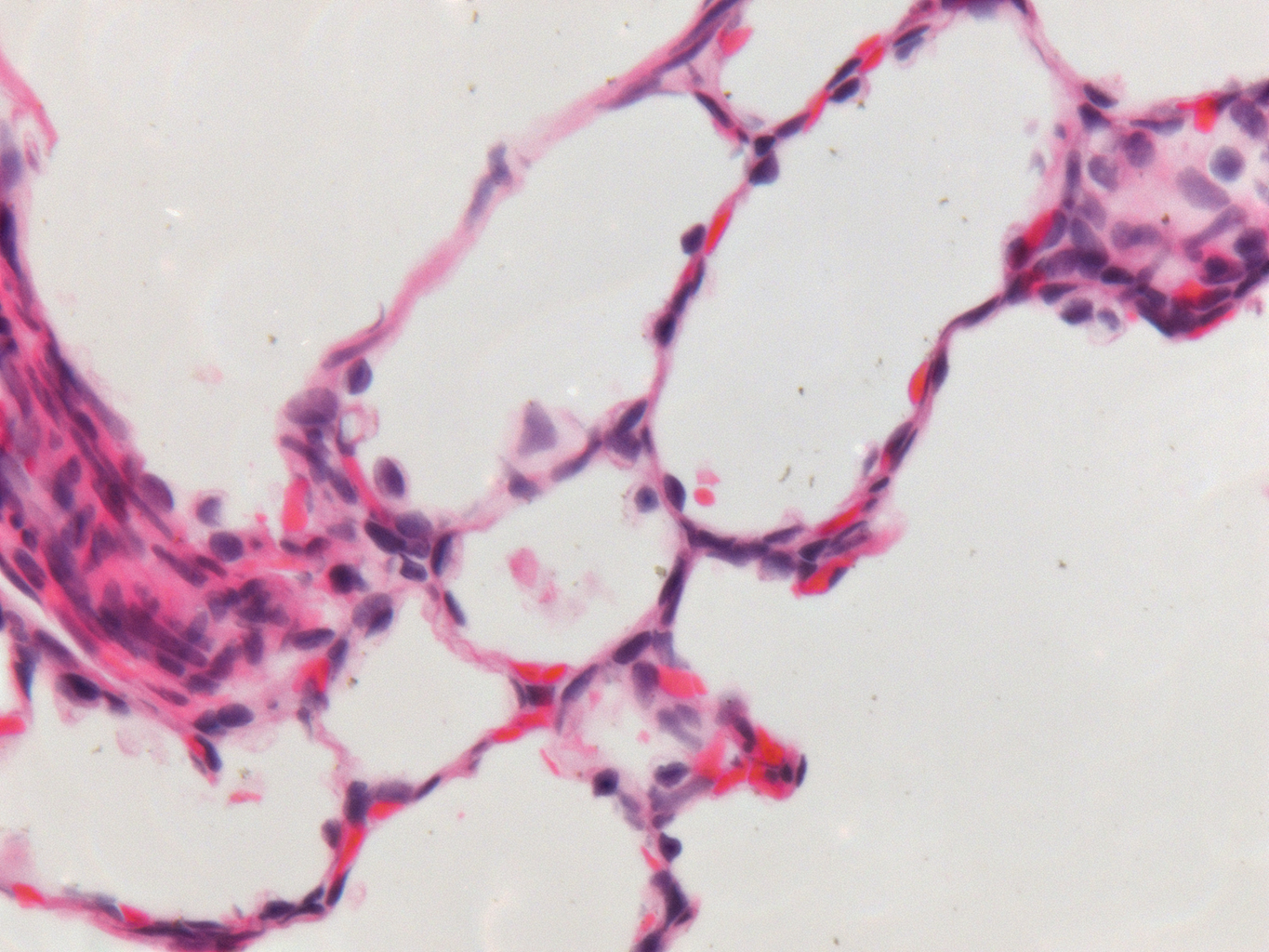
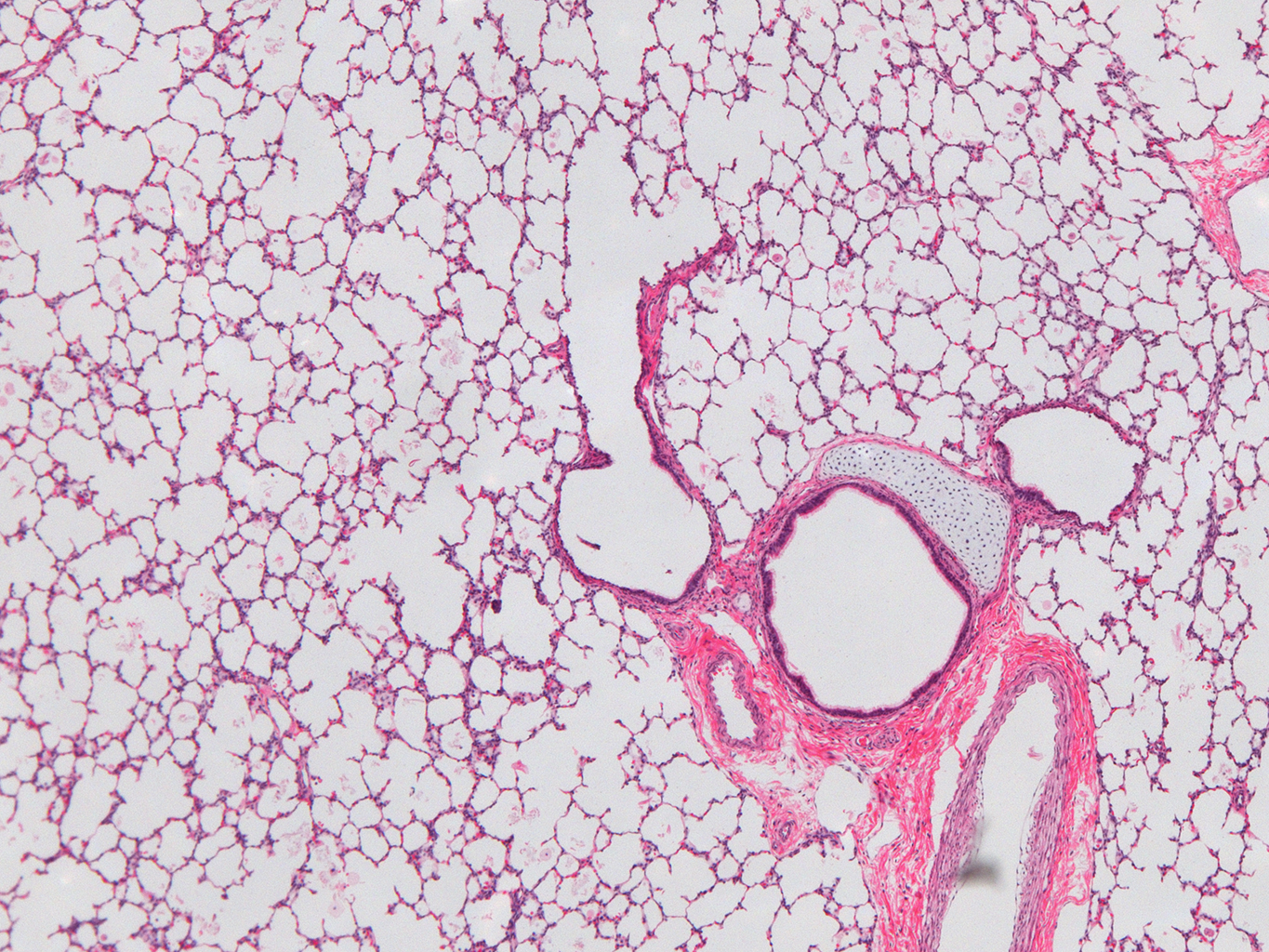
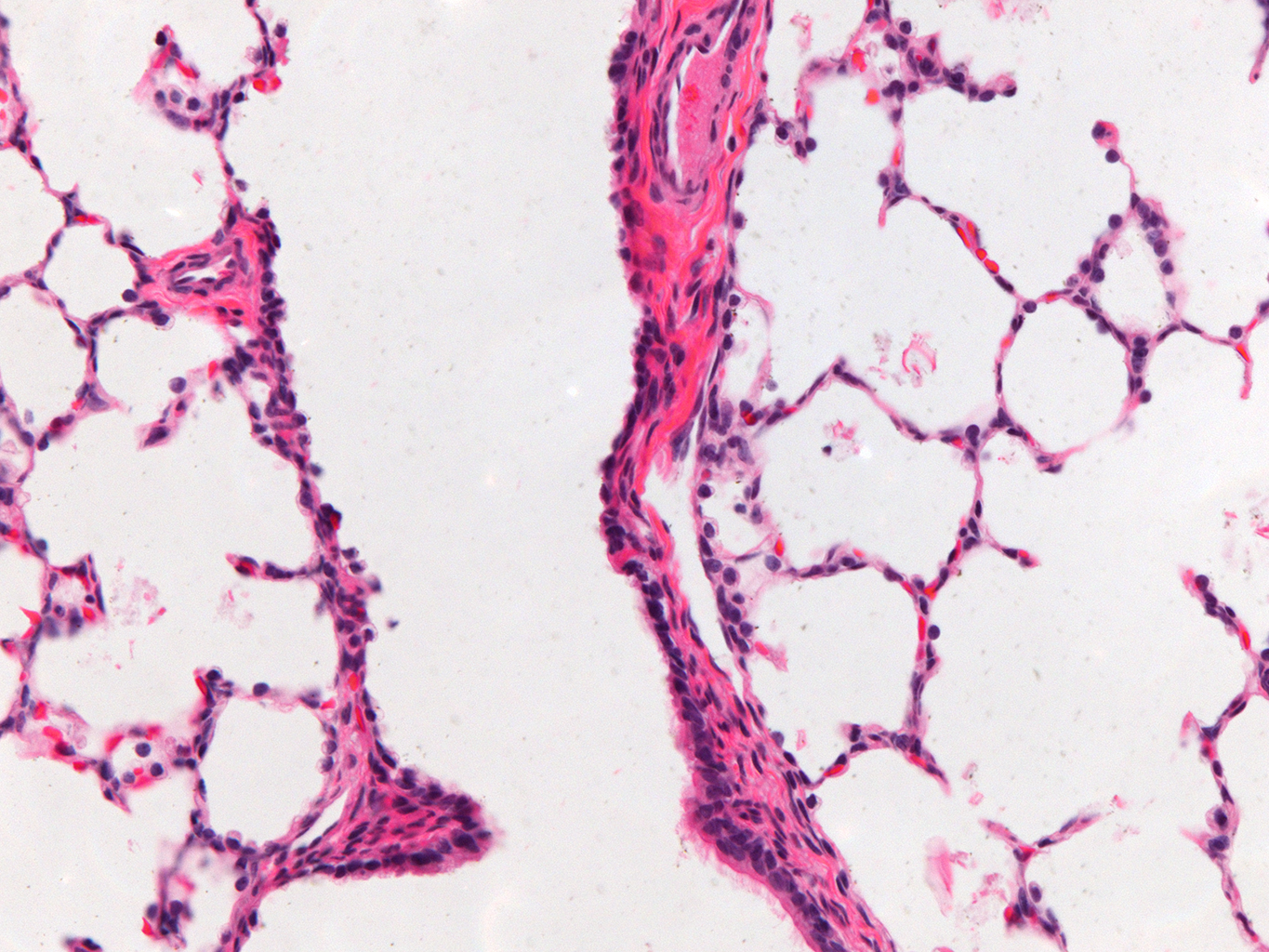
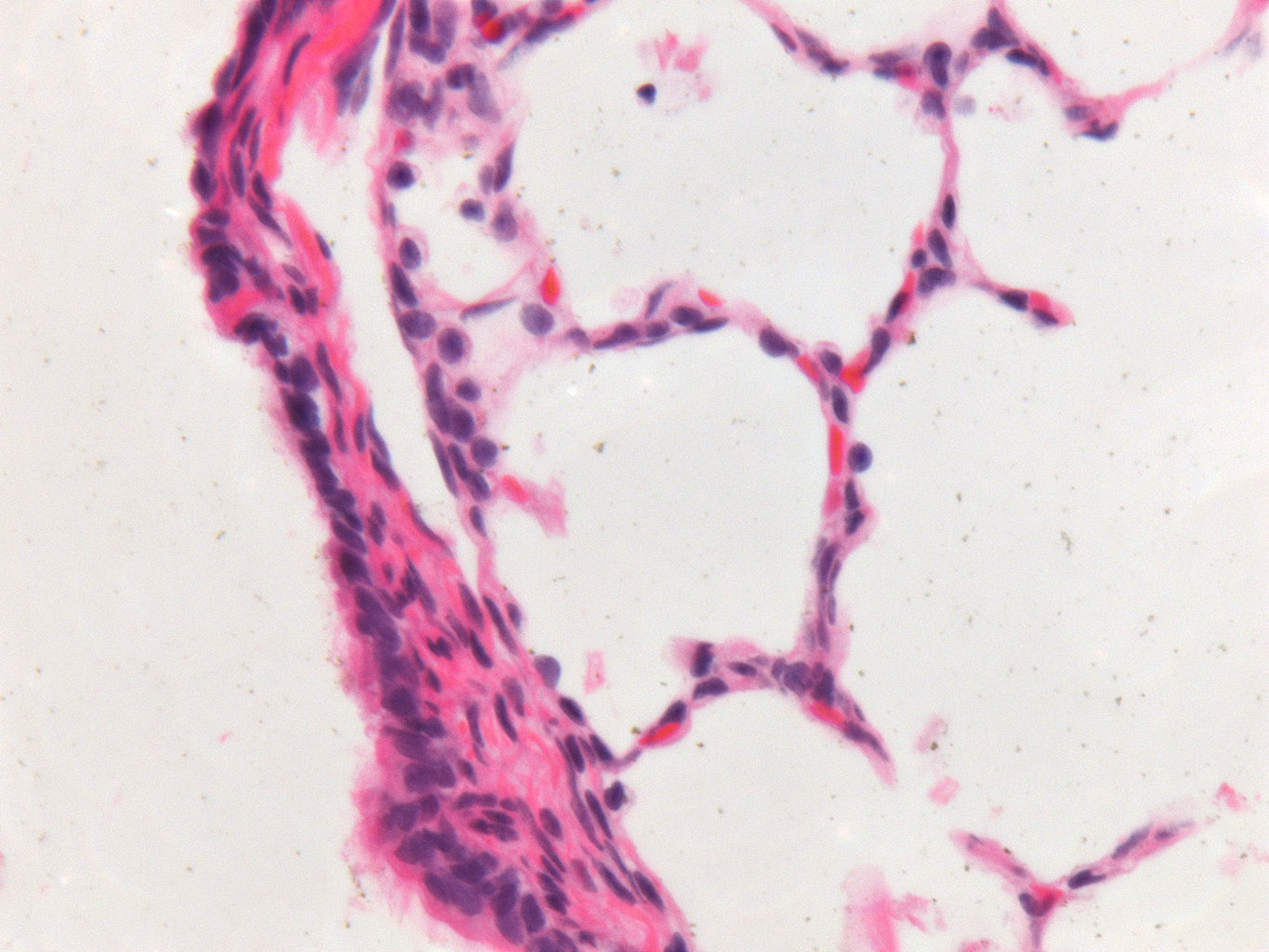
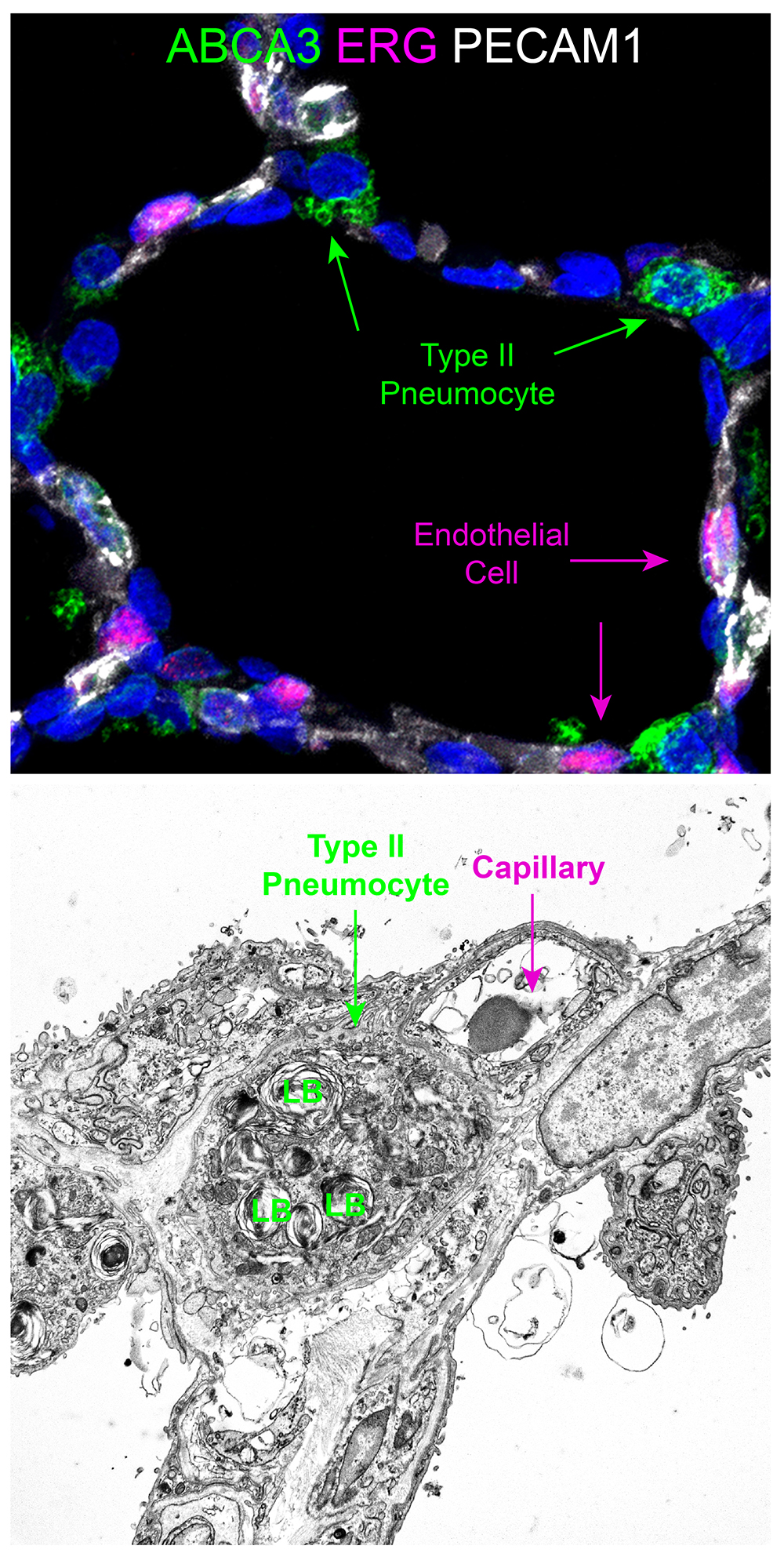
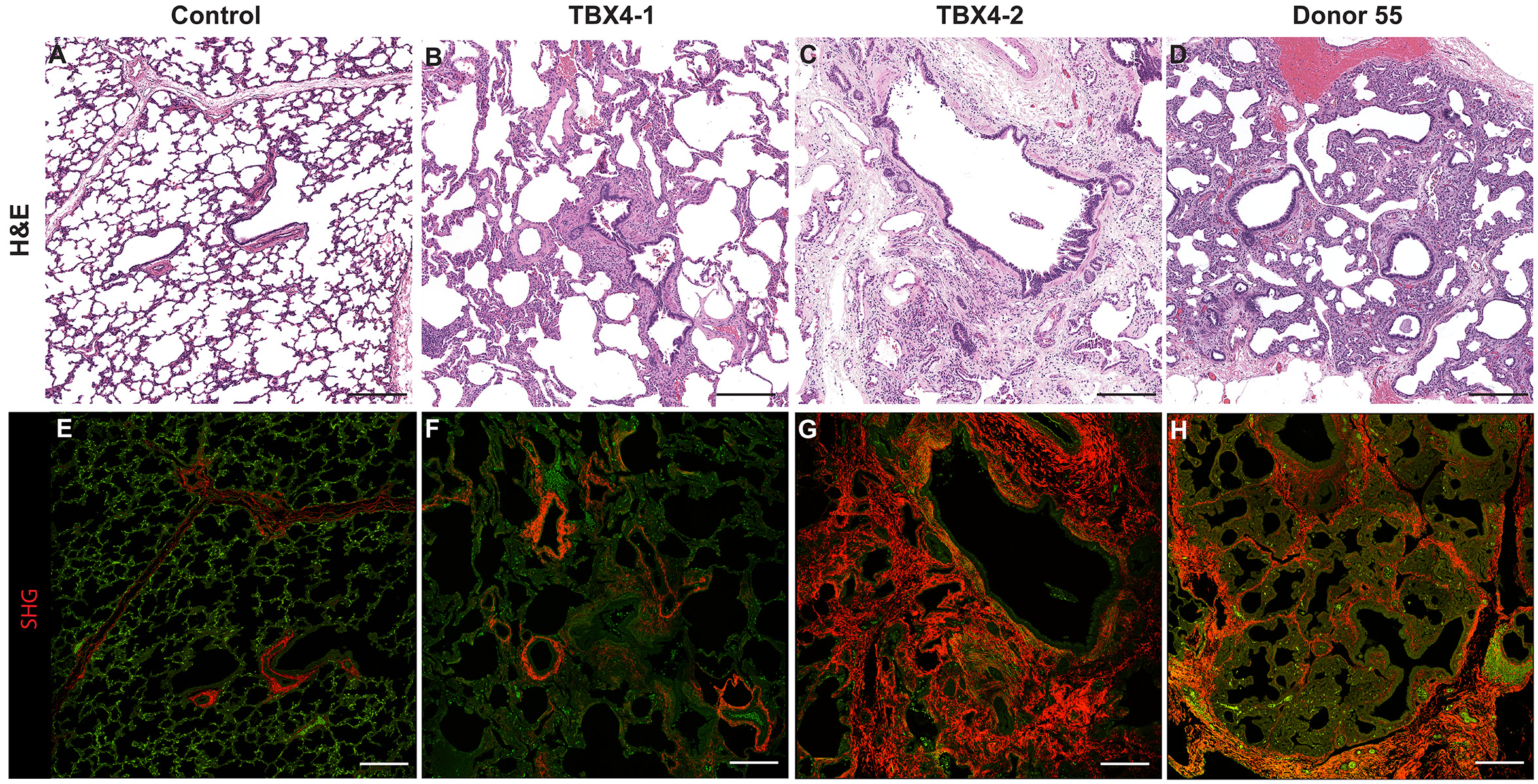
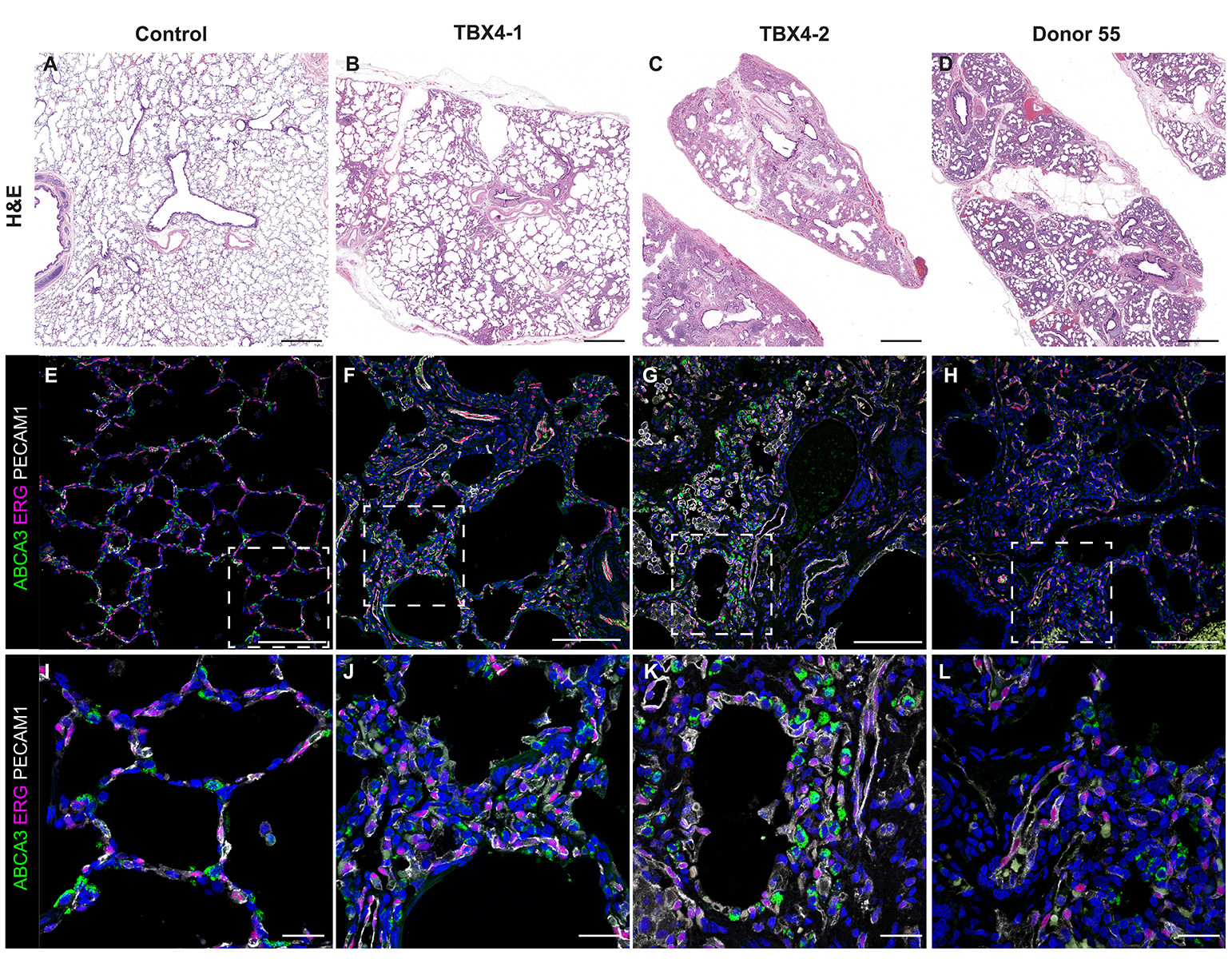
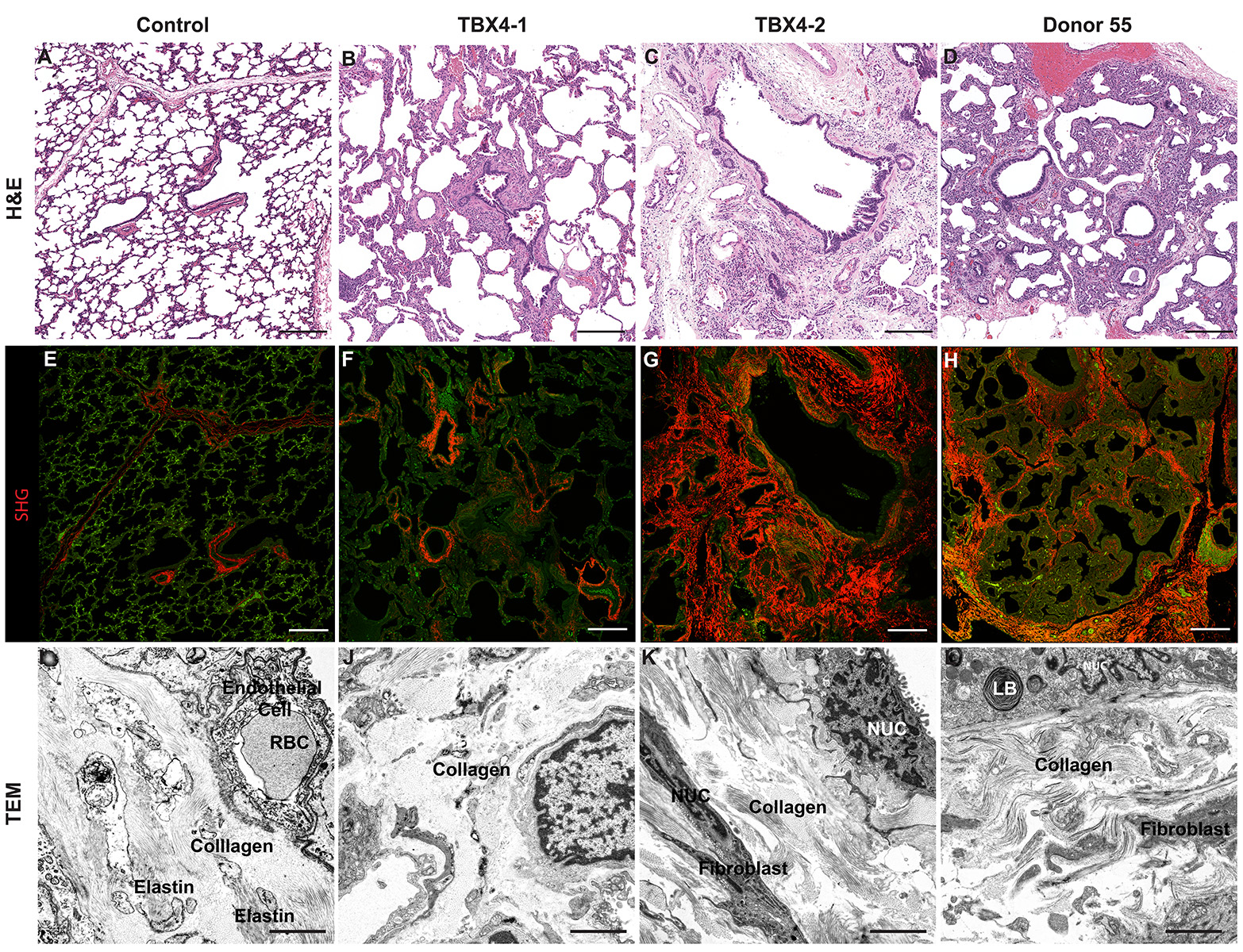
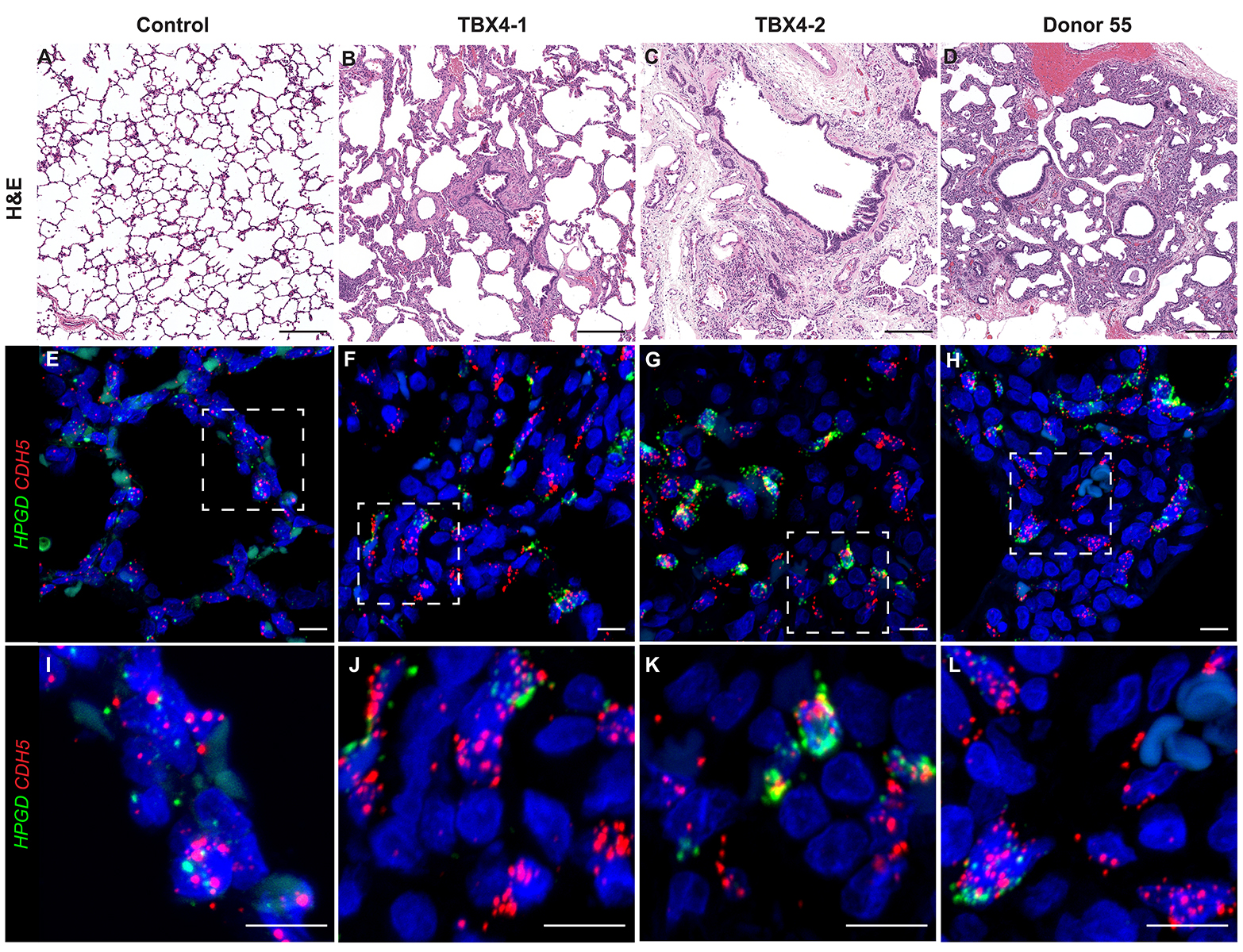
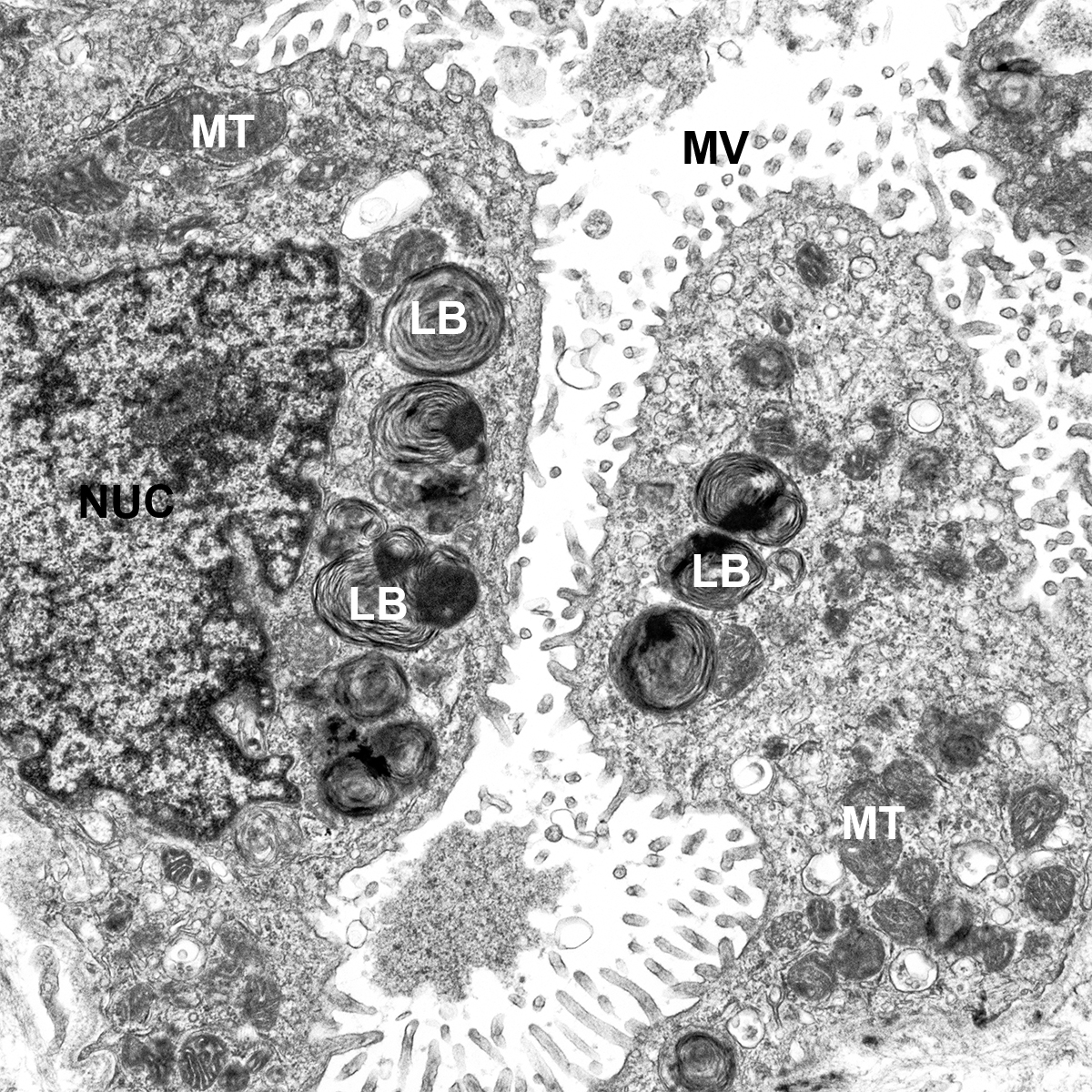
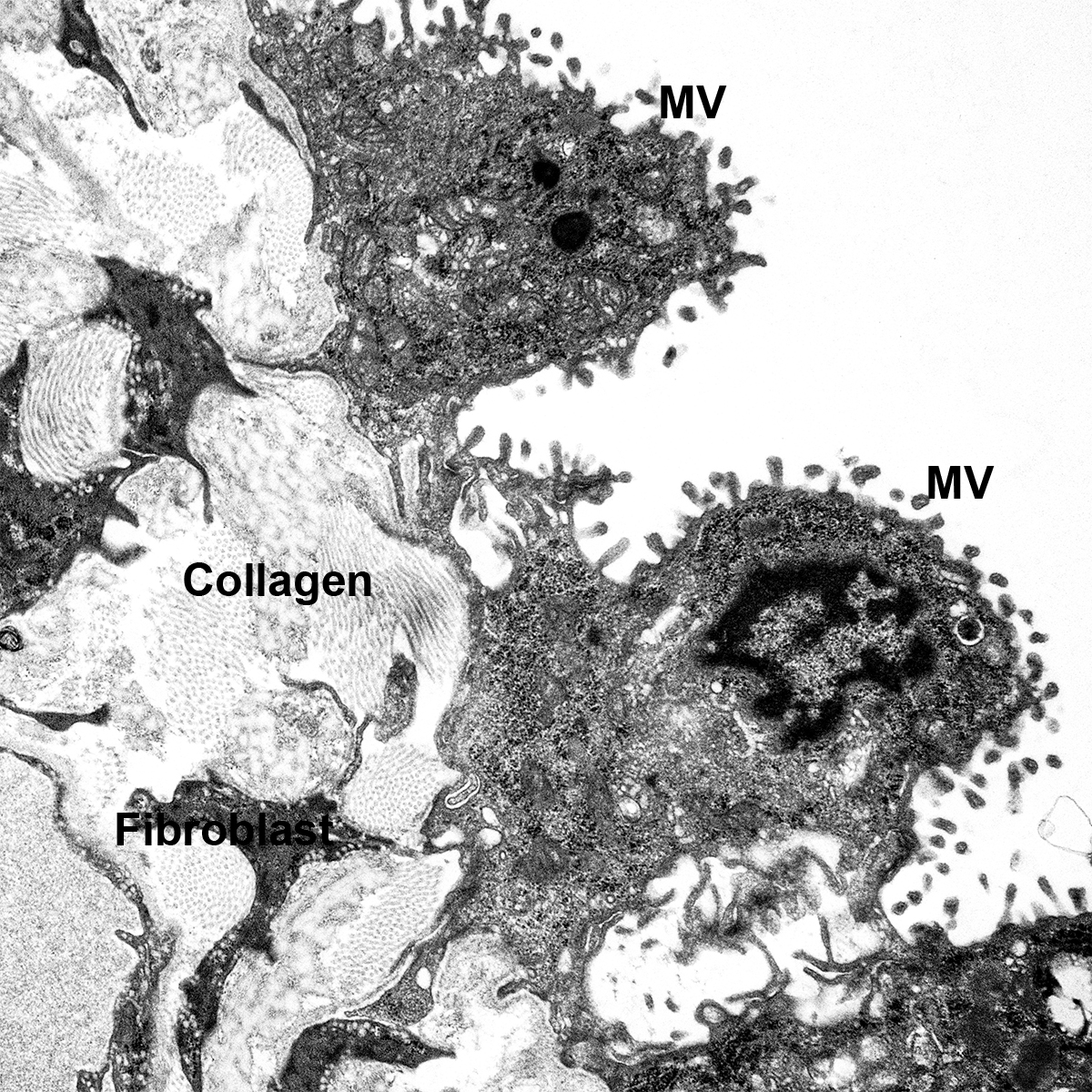
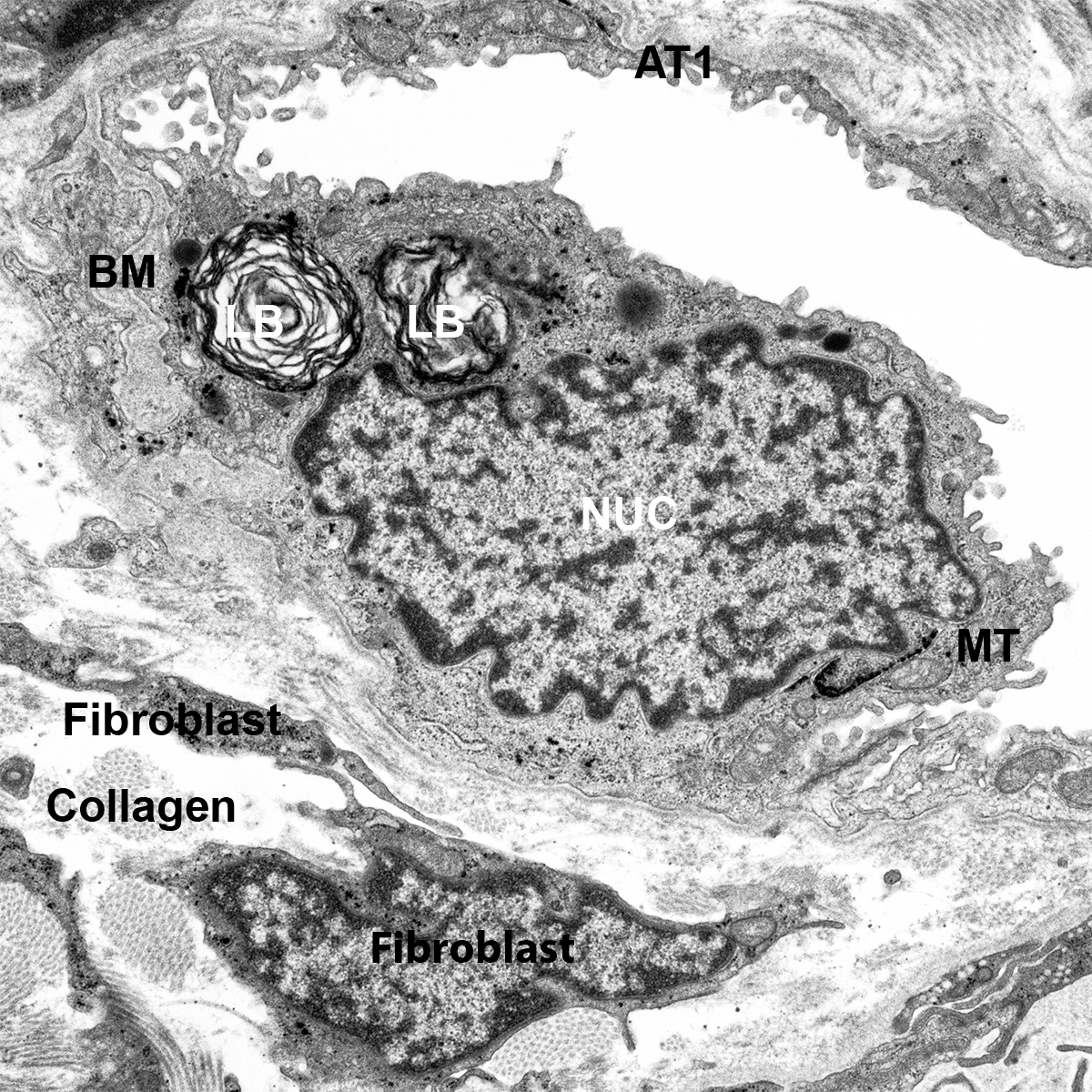
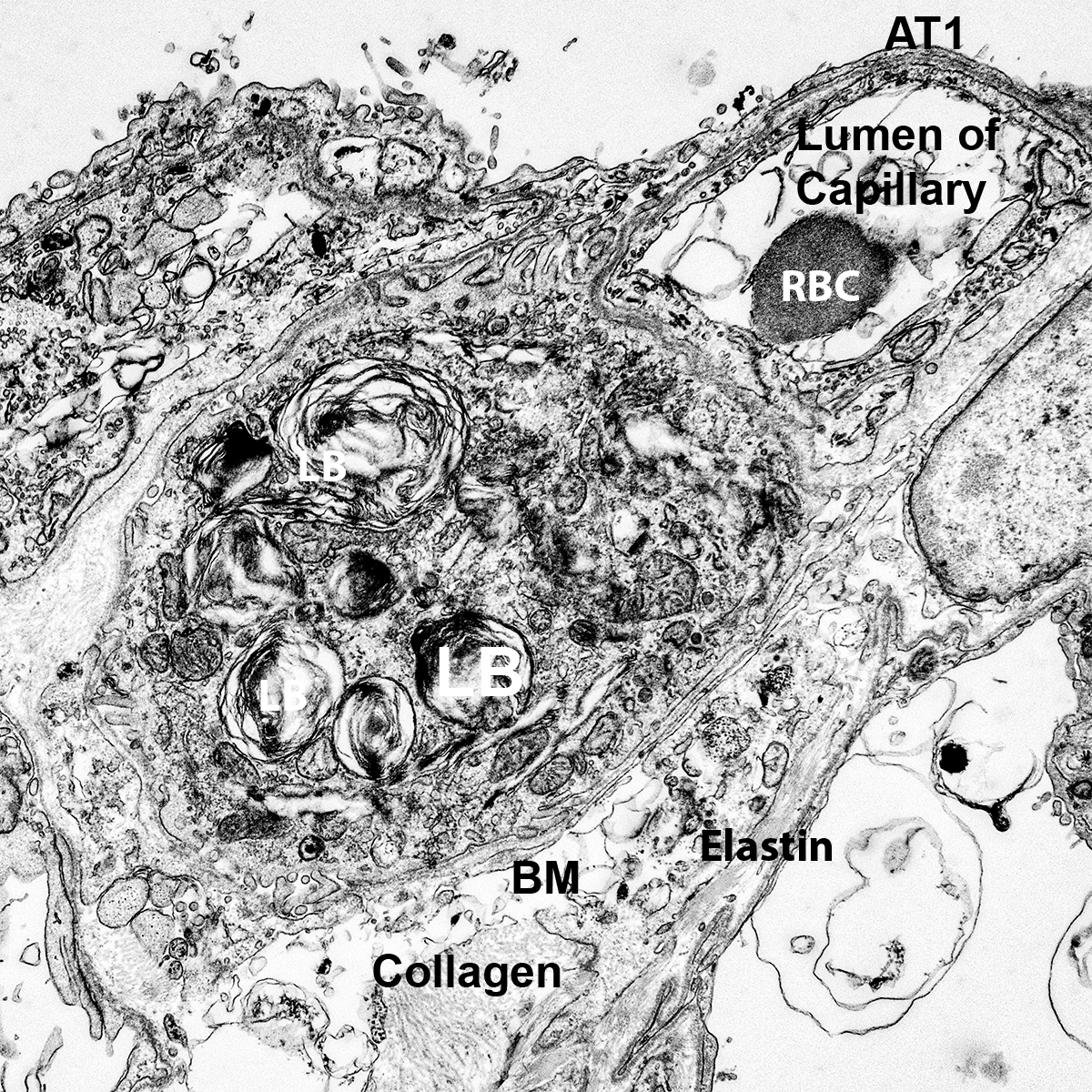
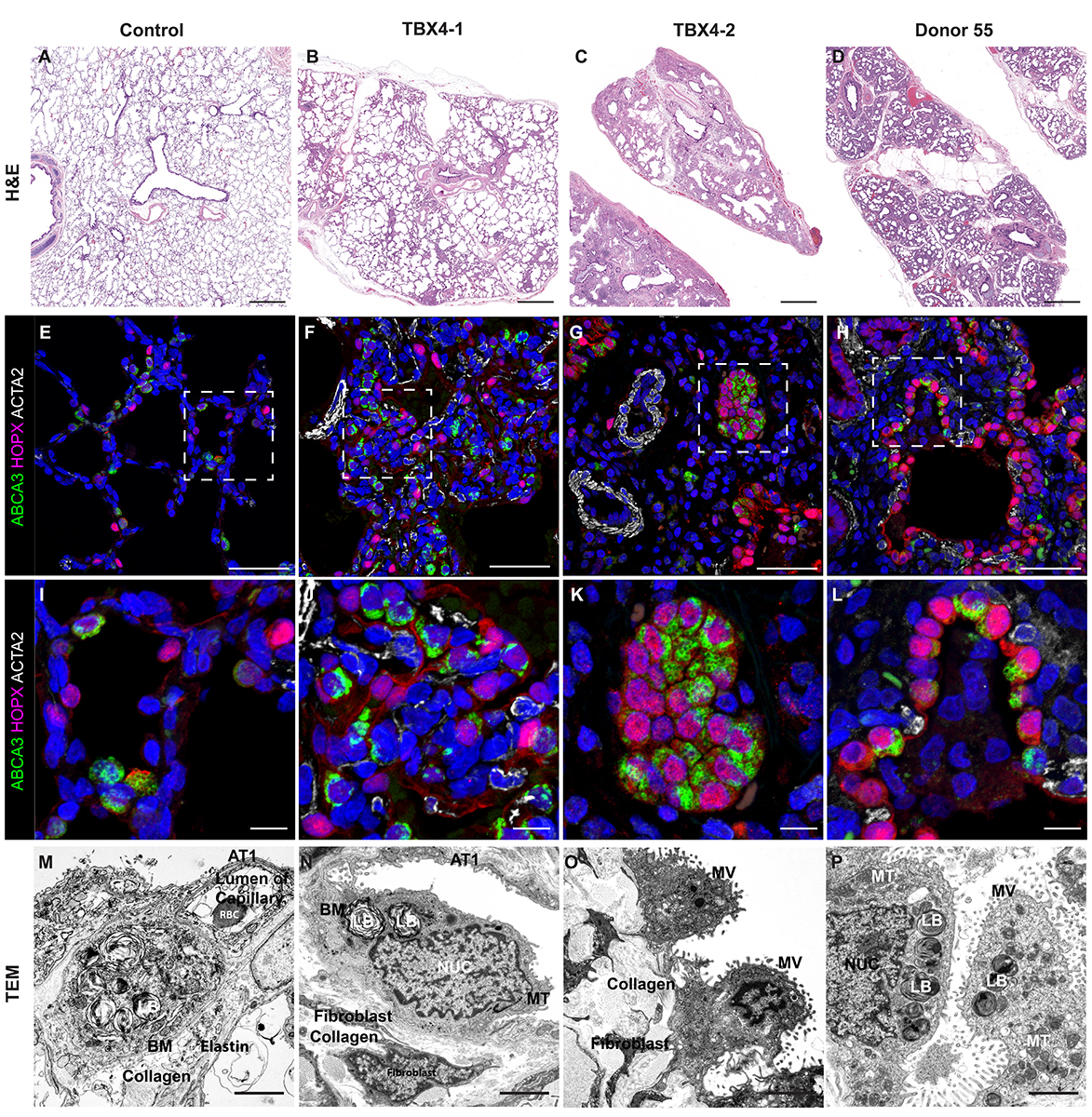
Donor Tissue Kindly Provided by Dr. Gloria Pryhuber from the University of Rochester Medical Center
- Paraffin sections were placed at 60oC overnight to melt paraffin.
- Paraffin sections were then placed in xylene (Xylenes Reagent Grade ACS Histology/Lab Grade Cat # 399000000 Pharma-AAPER) 3X for 10 minutes each.
- Rinsed slides in a series of 100% ETOH washes. 20 dips for the first wash, 3 minutes for the second wash, and 20 dips for the third wash.
- Rinsed slides in a series of 95% ETOH washes. 20 dips for the first wash, 3 minutes for the second wash, and 20 dips for the third wash.
- Rinsed slides in 70% ETOH for 3 min.
- Rinsed slides for 3 minutes in running dH2O.
- Slides were then incubated in 3% Glacial Acetic Acid (S101, Poly Scientific) for 3 minutes.
- Slides were then incubated in filtered Alcian Blue (S111A, Poly Scientific) for 30 minutes.
- Wash slides under cold running tap water for 10 minutes.
- Incubate for 2 minutes in Nuclear Fast Red (S248, Poly Scientific).
- Wash under flowing dH2O until excess Nuclear Fast Red is washed away.
- Incubated slides in 90% ETOH for 20 dips, repeat in different 90% ETOH.
- Incubated slides in 100% ETOH for 20 dips, repeat in different 100% ETOH.
- Washed slides in 2X in Xylene, followed by 6 more washes in xylene.
- Coverslip with Permount. (Permount Toulene Solution UN 1294, Cat # SP-15-500 Fisher Scientific).
Tissue Used:
LMR14.8.4.3