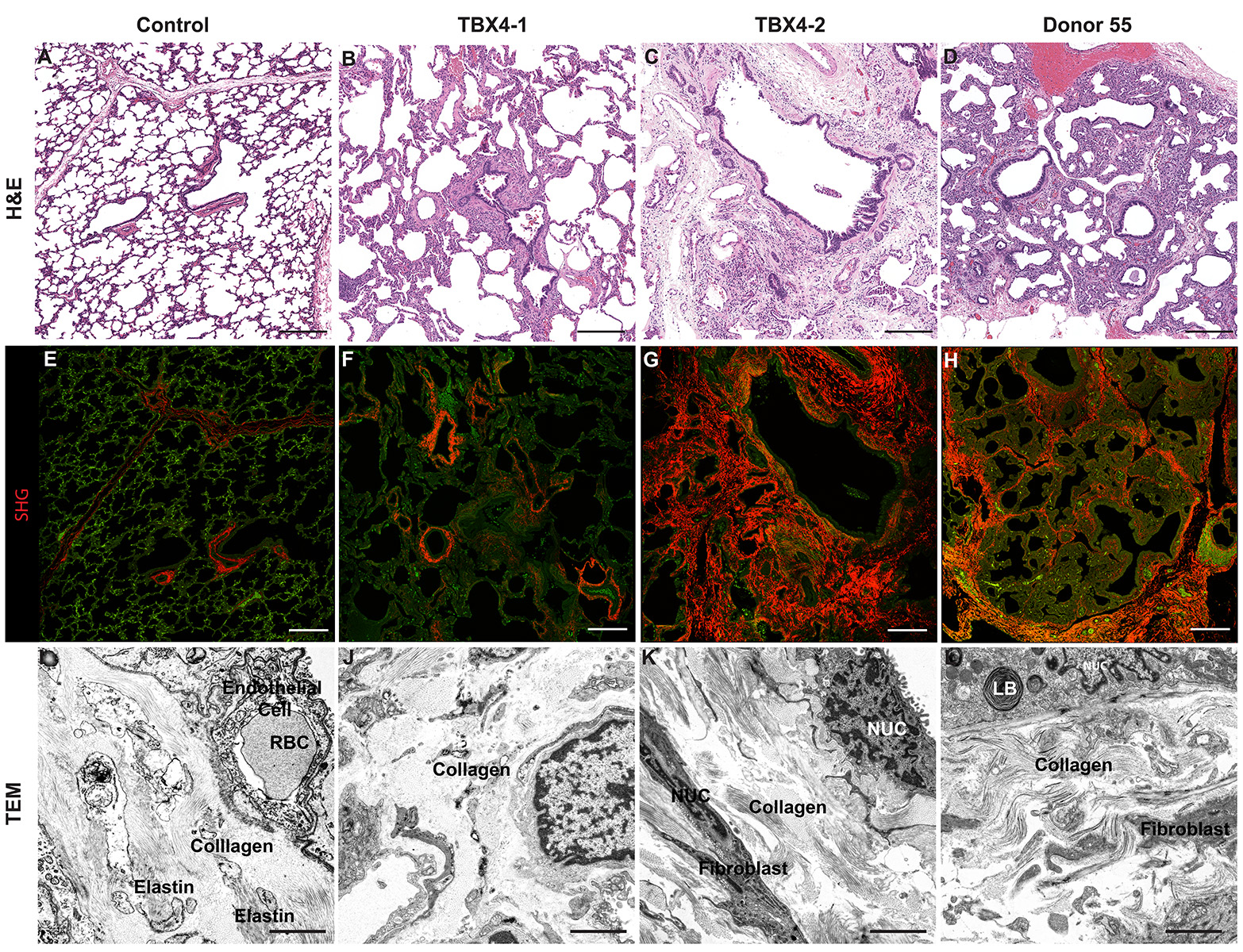
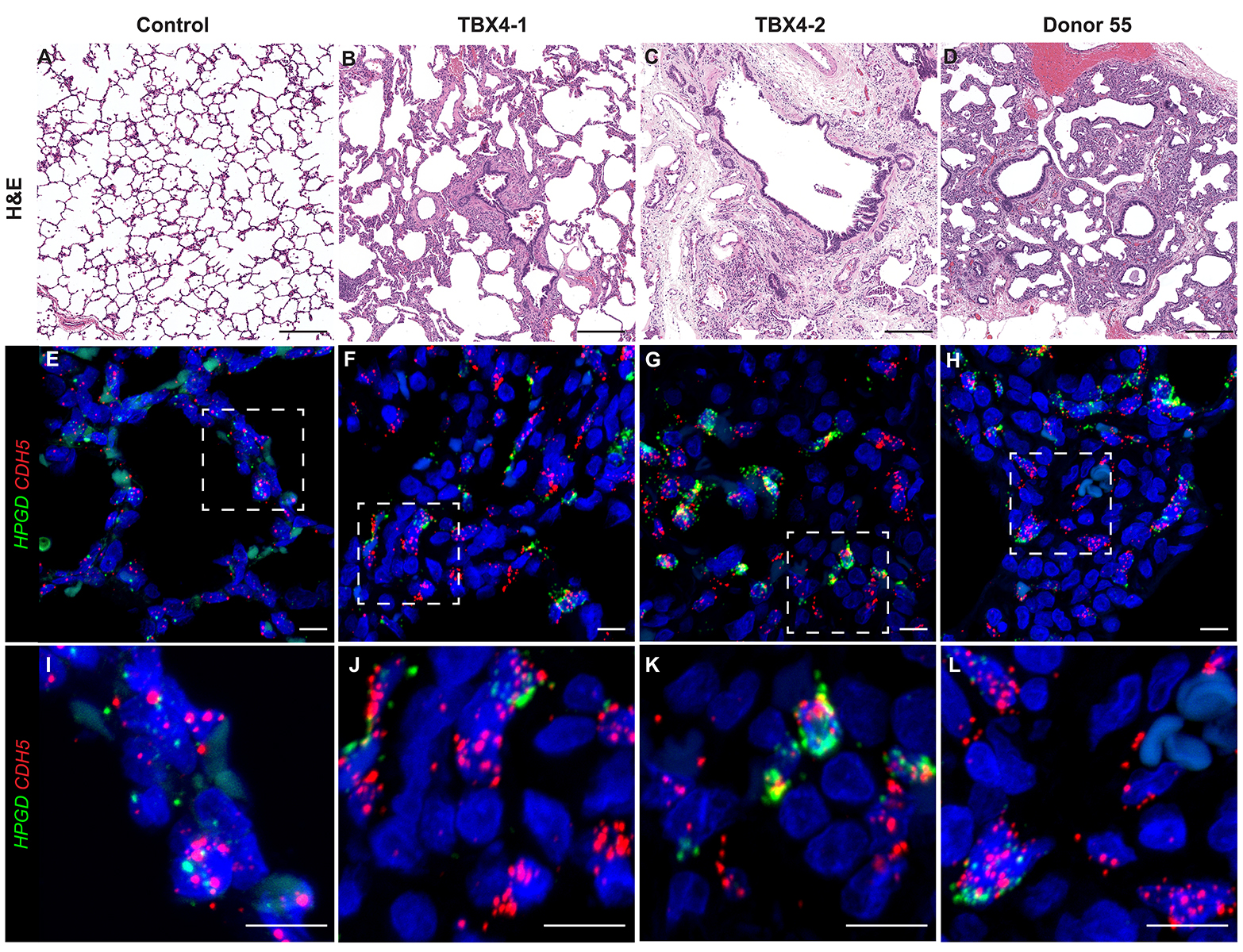
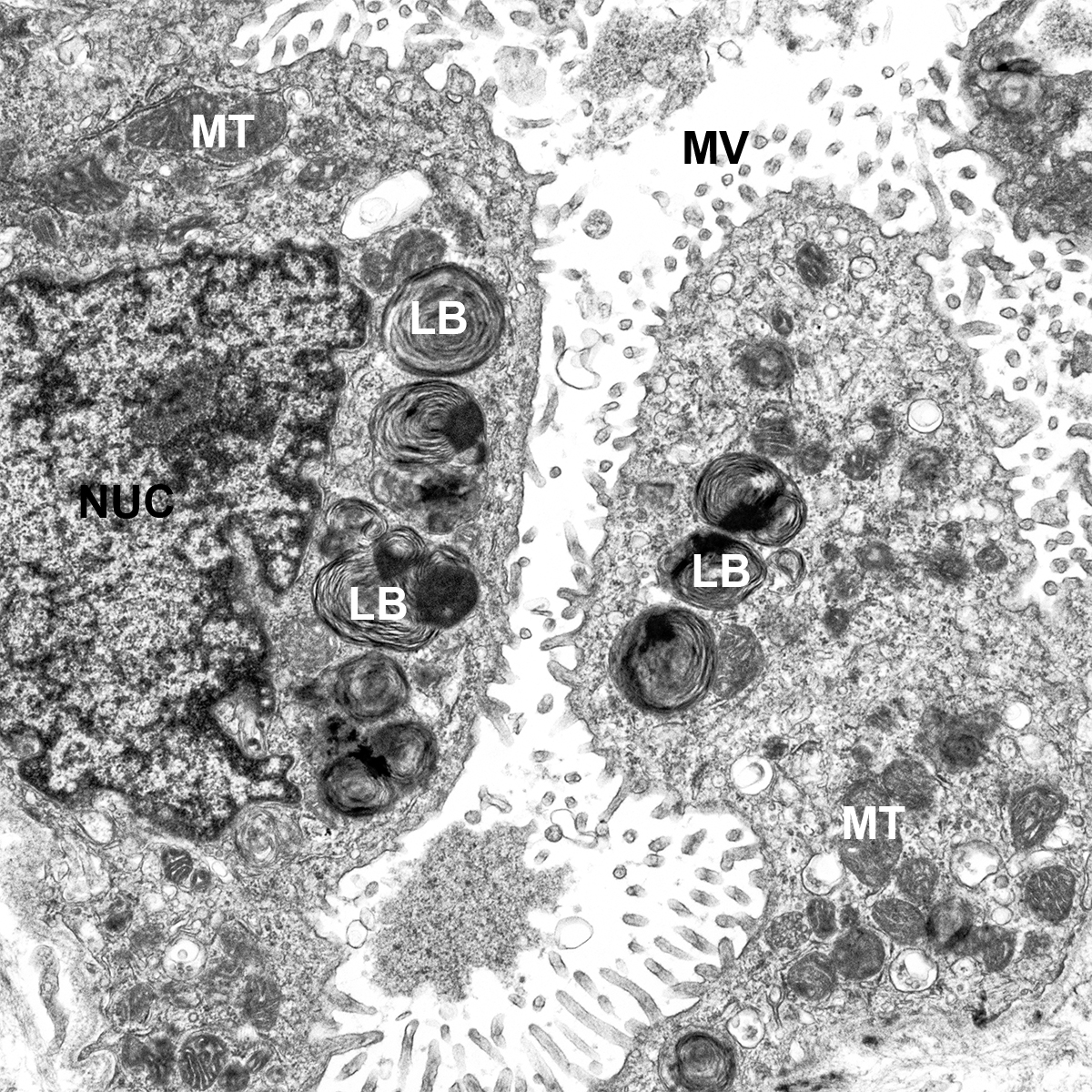
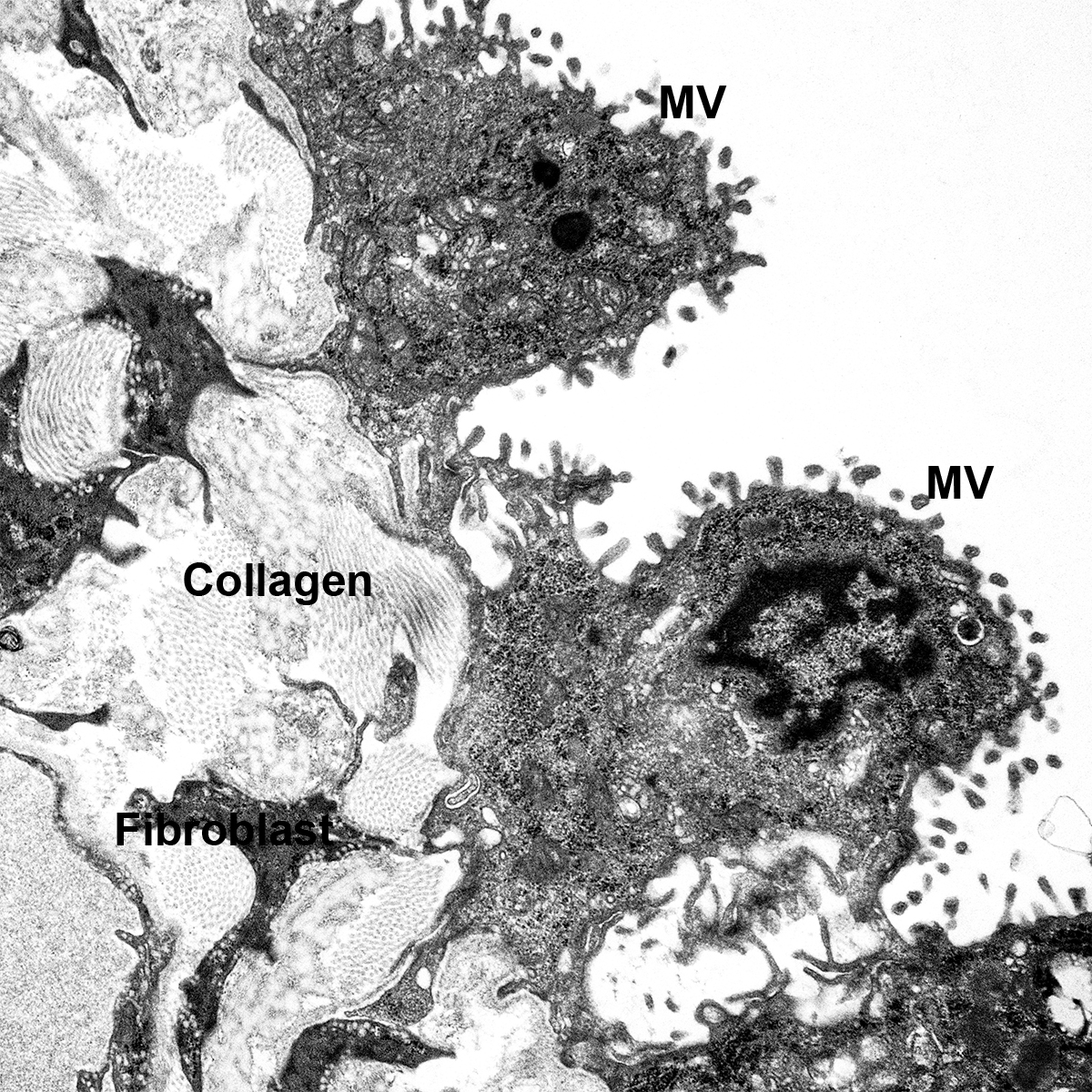
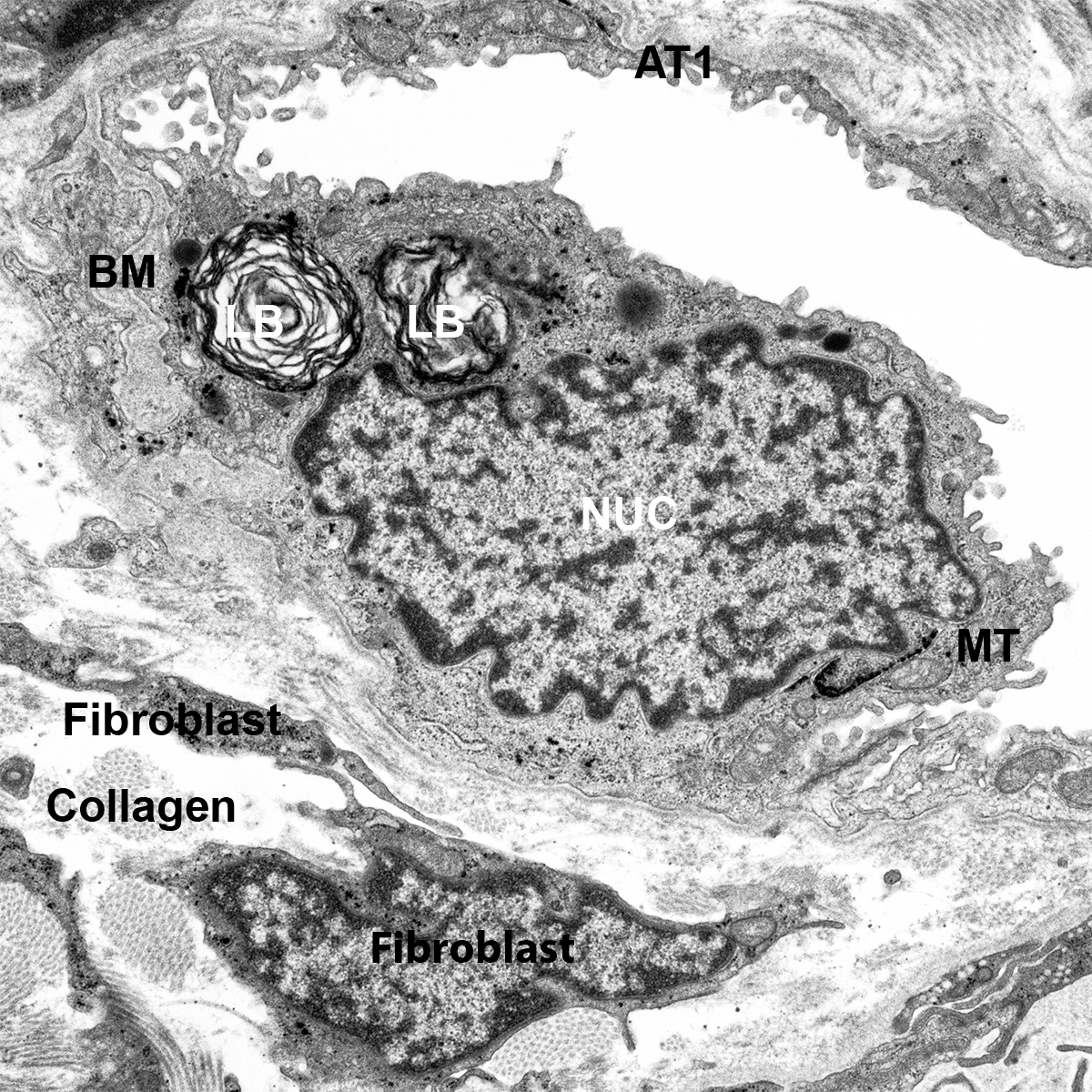
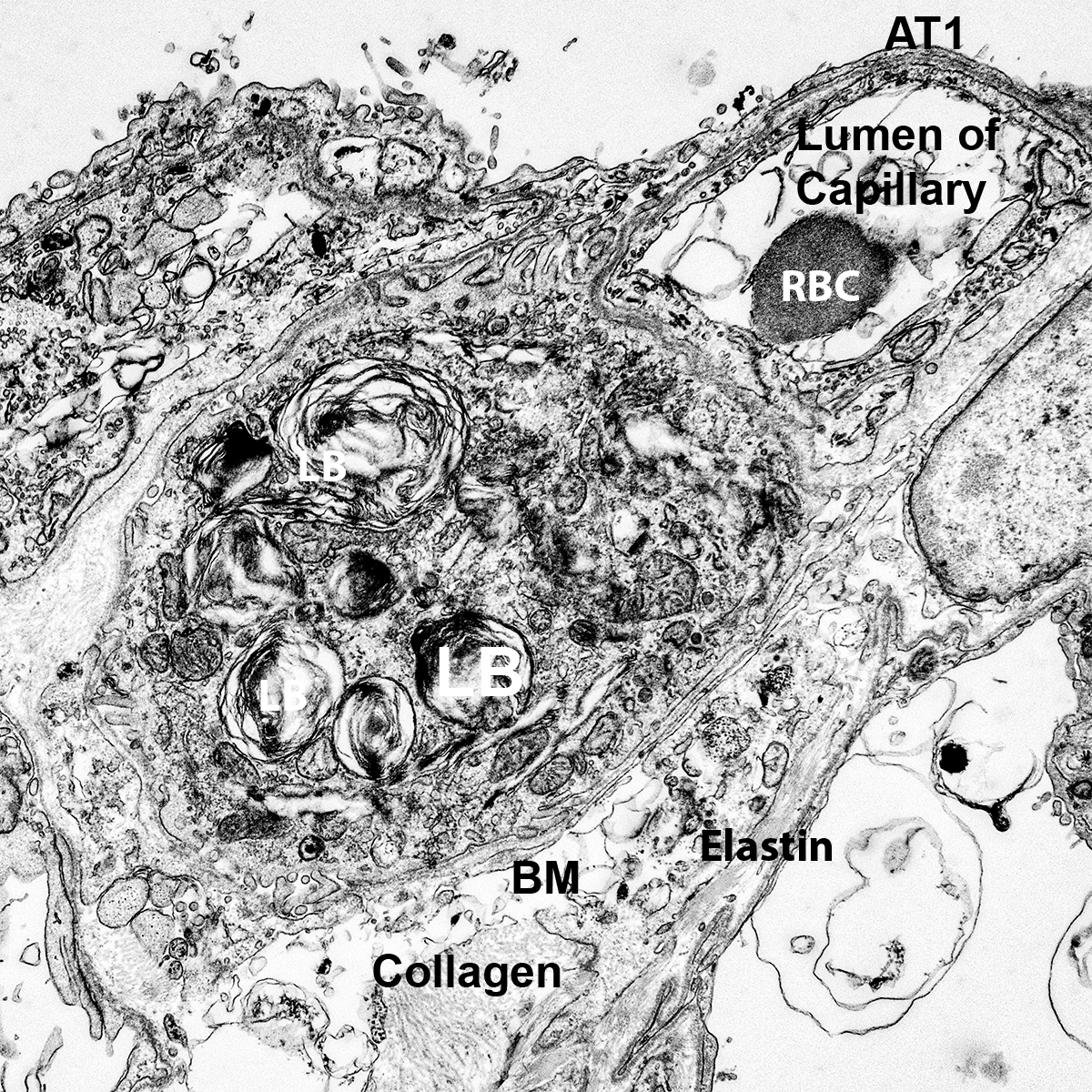
Dispase Lung Digestion
- Thaw the required volume of dispase on ice. For each mouse, add 1ml of dispase to a 14 mL round bottom polypropylene tube and place tubes into a 37oC heat block. Place the dispase needed for instilling into the lungs into an additional tube at 37o
- For each mouse prepare a C-tube by adding 5mL of DMEM containing 25 mM HEPES buffer and Penn/Strep.
- Anesthetize the mouse with 0.1mL of sodium pentobarbital. Secure animal for dissection. Spray mouse lightly with ethanol,
- Open the abdomen and move the organs to one side to expose the kidneys. Severe the renal artery and descending aorta to exsanguinate. Immediately raise the head of the animal. Using a strip of absorbent paper, cut to a point at both ends, remove as much blood as possible from the abdominal cavity.
- Open the skin from the abdomen to the top of the throat. Dissect away the salivary glands. Cut along the length of the sternum. Continue cutting to open the muscle tissue above the trachea. Remove the rib cage. Remove any muscle that obscures the trachea.
- Use curved forceps to pull a piece of size 2.0 suture under the trachea. Draw the ends into a loose single knot.
- Use a 20G, 0.75 inch angiocath catheter to cannulate the trachea. Remove the protective caps, so that just the needle and soft sheath remain. Insert the catheter into the trachea just above the cartilaginous ring, using the needle to penetrate. Stop when the needle is still visible at the bottom of the trachea.
- Support the catheter with one hand and carefully slide the needle out with the other hand. Have a transfer pipette available. It can be placed under the catheter to steady, if needed.
- Check the position of the catheter in the trachea, and tighten the suture, taking care that the catheter does not slide out as the knot is tightened.
- Fill a 10 mL syringe with saline and attach a 21G 1.5 inch needle. With forceps hold the tip of the heart and lift it up to 90o. Locate the pulmonary artery, running vertically down the back side of the heart and off center to the right side. Hold the needle straight up and down. Starting about ¼ of the way down from the top, insert the needle into the heart behind the line of the pulmonary artery. Direct the needle so that the tip just emerges into the right auricle. Perfuse lung with 10 mL of saline. If the position of the needle is correct, the RBCs will be flushed from the lungs, leaving the lungs white. The lung can be perfused a second time if needed.
- Fill a 3mL luer lock syringe with dispase that has been warmed to 37oC and attach it to the cannula. Before instilling the dispase, check that the tip of the catheter is visible, as it can be pushed deeper as the syringe is attached. Instill dispase to fully inflate the lung (2.3 mL for post natal day 28 mice and 3 mL for 6-8 week old mice).
- Tighten the suture knot to close the trachea. Be careful that the catheter does not slide out, as it is easily pushed out as the knot is closed.
- The lungs will be fragile from the instilled dispase. Dissect out the lung as gentle as possible, with the goal of keeping the lung and trachea intact with the thymus and heart attached. Transfer the dissected lung/trachea/thymus/heart complex into a 14 mL polypropylene tube containing 1 mL dispase. Incubate at 37oC for 6 minutes.
- Pour the dispase from the polypropylene tube. In a dish containing room temperature saline, excise the lung from the heart and thymus. Separate the individual lobes from the lung. Remove any visible airway tissue and transfer the lobes to a prepared C-tube. Hold at room temperature. Prepare 4-8 lungs to this point, and then continue processing them through the remaining steps.
- Add 60 μL of DNAse (10 KU/mL) to the C-tube. Insert C-tube into the GentleMACs Dissociator, select program m_lung_1.01, and press start. The program dissociates the lung in 10 seconds. Re-run if the tissue is not fully dissociated.
- Incubate lungs at room temperature for 10 minutes.
Cell Isolation
- Centrifuge C-tube at 1500 rpm (433 x g) for 6 minutes at 4o Remove supernatant and combine cells from two mice cell digestions in 2 mL of red cell lysis buffer and incubate on ice for 2 minutes.
- Add 8 ml of DMEM media and centrifuge at 1500 rpm for 6 minutes at 4o
- Remove supernatant and resuspend in 10ml DMEM and filter cells through 30 μM cell strainer. Wash C-tube with 5 mL of DMEM media and pour onto cell strainer.
- Count cells – use a 1:10 dilution. Typical yield: 40-50 million cells per mouse.
- For four mice use two columns for CD45+
- Centrifuge the conical tubes at 1500 rpm for 6 minutes at 4o Resuspend 107 cells in 90 μL of MACs buffer and add 10 μL CD16/32 to block Fc receptors. Incubate for 10 minutes at 4oC.
(For higher cell numbers, scale up buffer and antibody volume accordingly)
- Then add 10 μL anti-mouse CD45 microbeads per 107 Mix well and incubate at 4°C for 15 minutes.
(For higher cell numbers, scale up buffer and antibody volume accordingly)
- Wash cells by adding 1−2 mL of MACs buffer per 10⁷ cells and centrifuge at 1500 rpm for 6 minutes at 4oC. Aspirate supernatant completely.
- Resuspend up to 10⁸ cells in 500 μL of MACs
- Place LS Column in the magnetic field of a MACS Separator (orange magnet attached to stand).
- Pre-rinse the column with 3 mL of MACs buffer and place 15 mL conical under columns to collect eluent.
- Apply the cell suspension (500 μL) onto the pre-separator filter over the rinsed column.
- Proceed with three washing steps with 3 mL of MACs buffer each wash and add MACs buffer only after the previous wash has eluted through the column.
- Remove the column and collect the cells with 5mL MACs buffer in a 15 mL collection tube by firmly pushing the plunger into the column and keep as CD45+ cells.
- Centrifuge the CD45+ cells at 1500 rpm (433 x g) for 6 minutes at 4o Resuspend cells at a pre-determined concentration and do FACS to determine purity using 1.5 X 105 cells.
- Collect total effluent; this is the CD45– fraction and proceed with enrichment of CD326 (EpCAM)+ cells .
- Repeat the steps from 23 to 31 with CD326, CD31, and CD140α Microbeads to separate CD326+, CD31+, and CD140α + cells, respectively. At this point it will be possible to combine the preps and use one column.
- Centrifuge the CD45+, CD326+, CD31+, and CD140α + cell suspensions, resuspend in complete media (DMEM) and count the cells.
Reagents
DMEM media: Remove 35 mL from 1L bottle of sterile water. Add powdered media, sodium bicarbonate (3.7 g), 1M HEPES (25 mL) and PenStrep (10 mL). Mix to dissolve and filter.
DNase (Sigma D4527): Add 28 mL of sterile saline per vial. Aliquot 500 μL per tube and store at -20°C.
Corning (BD Biosciences, BD354235)
Red Cell Lysis Buffer (Cat# 00-4333-57, eBiosciences)
MACS BSA Stock Solution (Cat # 130‑091‑376, Miltenyi Biotec)
AutoMACS Rinsing Solution (Cat # 130-091-222, Miltenyi Biotec)
LS Columns (Cat# 130-042-401, Miltenyi Biotec)
FcR Blocking Reagent, mouse (Cat # 130-092-575, Miltenyi Biotec)
CD45 MicroBeads, mouse (Cat # 130 052 301, Miltenyi Biotec)
CD326 (EpCAM) MicroBeads, mouse (Cat # 130 105 958, Miltenyi Biotec )
CD31 MicroBeads, mouse (Cat # 130-097-418, Miltenyi Biotec )
CD140α MicroBeads, mouse (Cat # 130-101-502)
Pre-Separation Filters (30 µm) (Cat # 130-041-407, Miltenyi Biotec )
Ethylenediaminetetraacetic Acid (Cat # BP2482-100, Fisher Scientific)
***this protocol is adapted from Messier et al., Experimental Lung Research 2012.