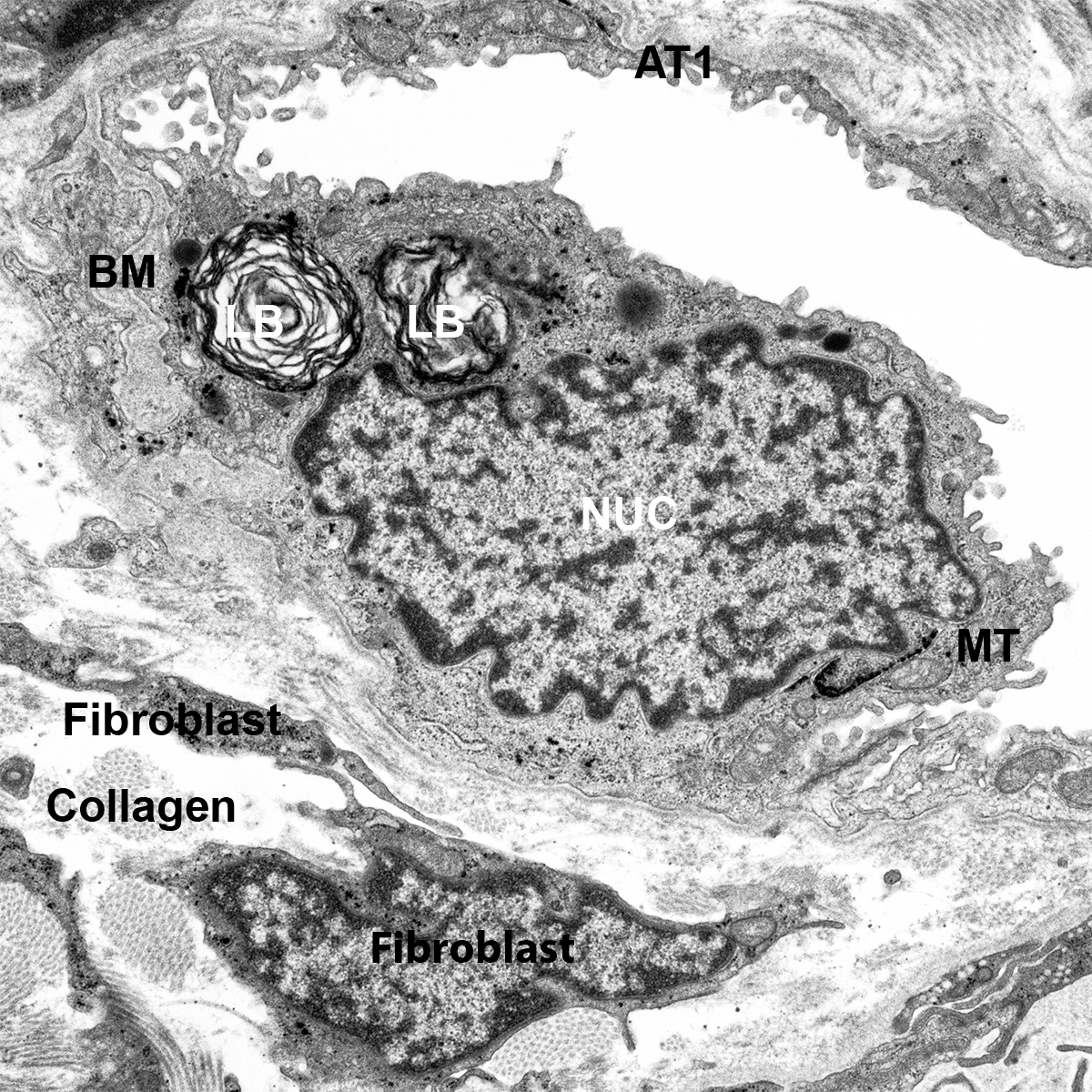
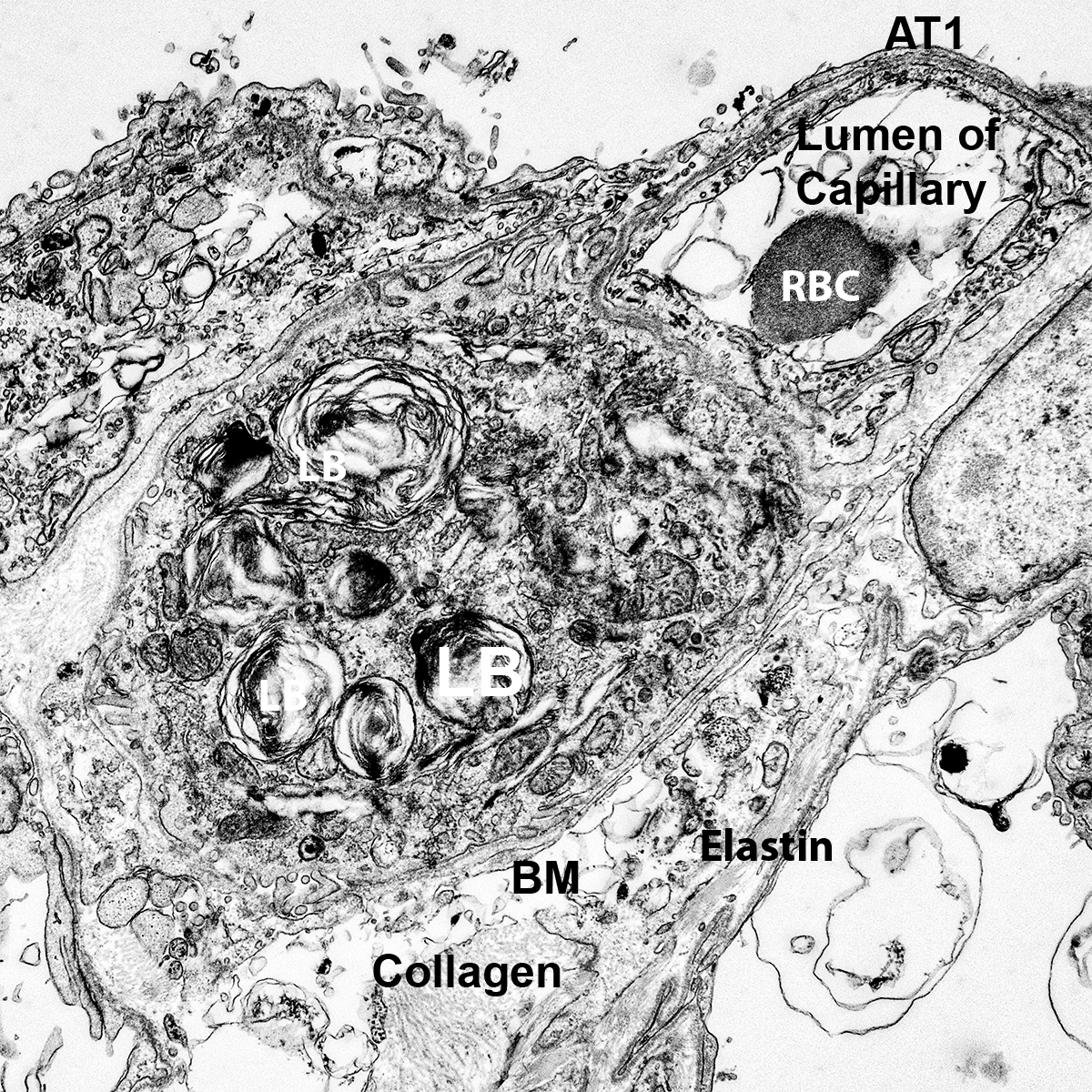
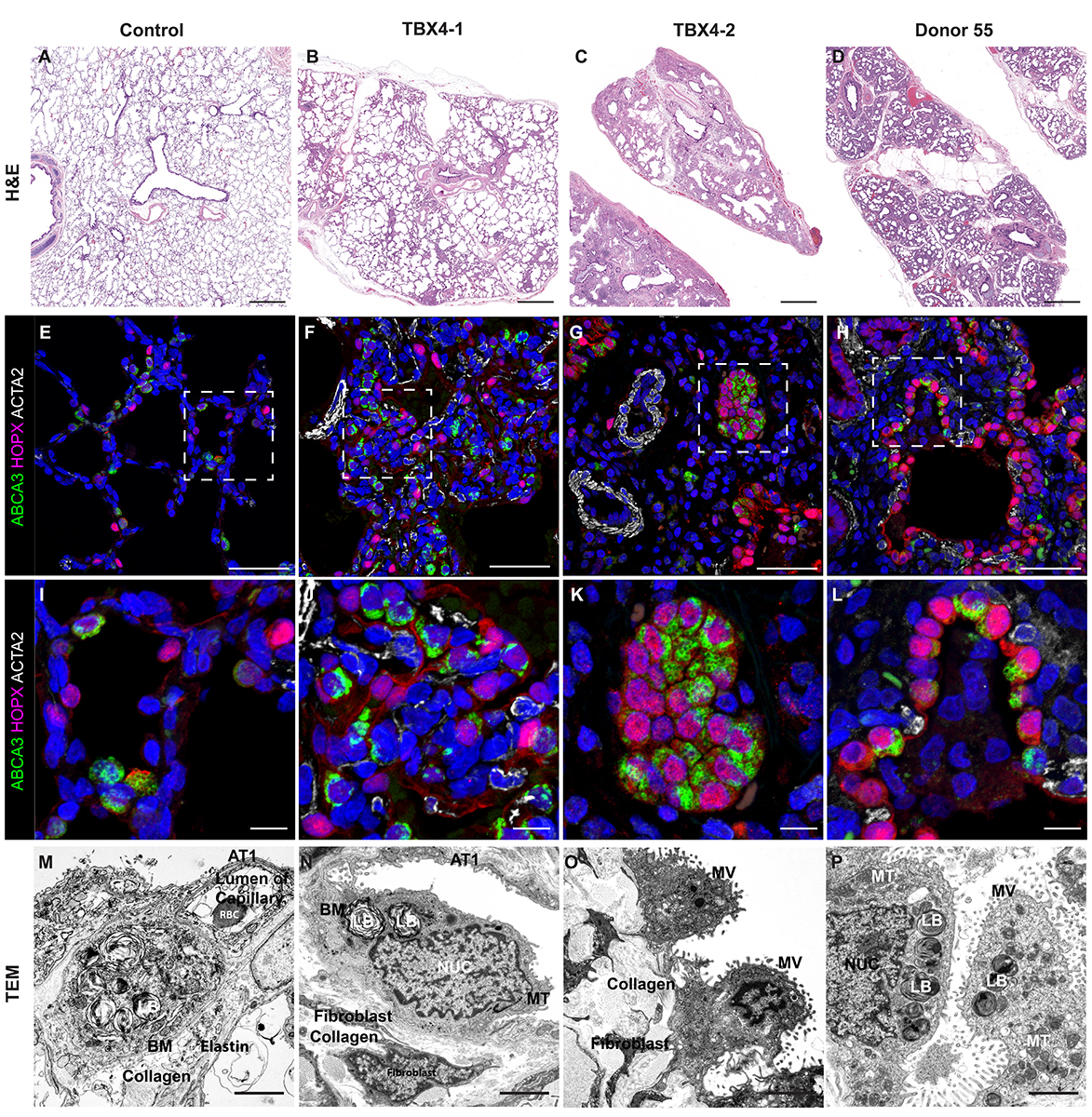
- Cut 7 µm sections of frozen tissue on a cryostat and dry overnight at room temperature. Alternatively, 5 µm sections of paraffin embedded tissue can be used.
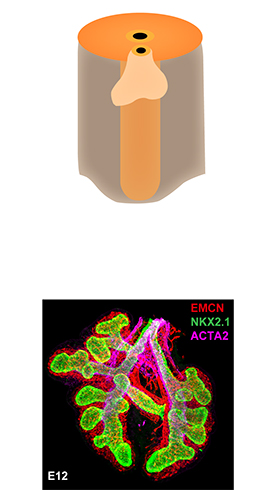
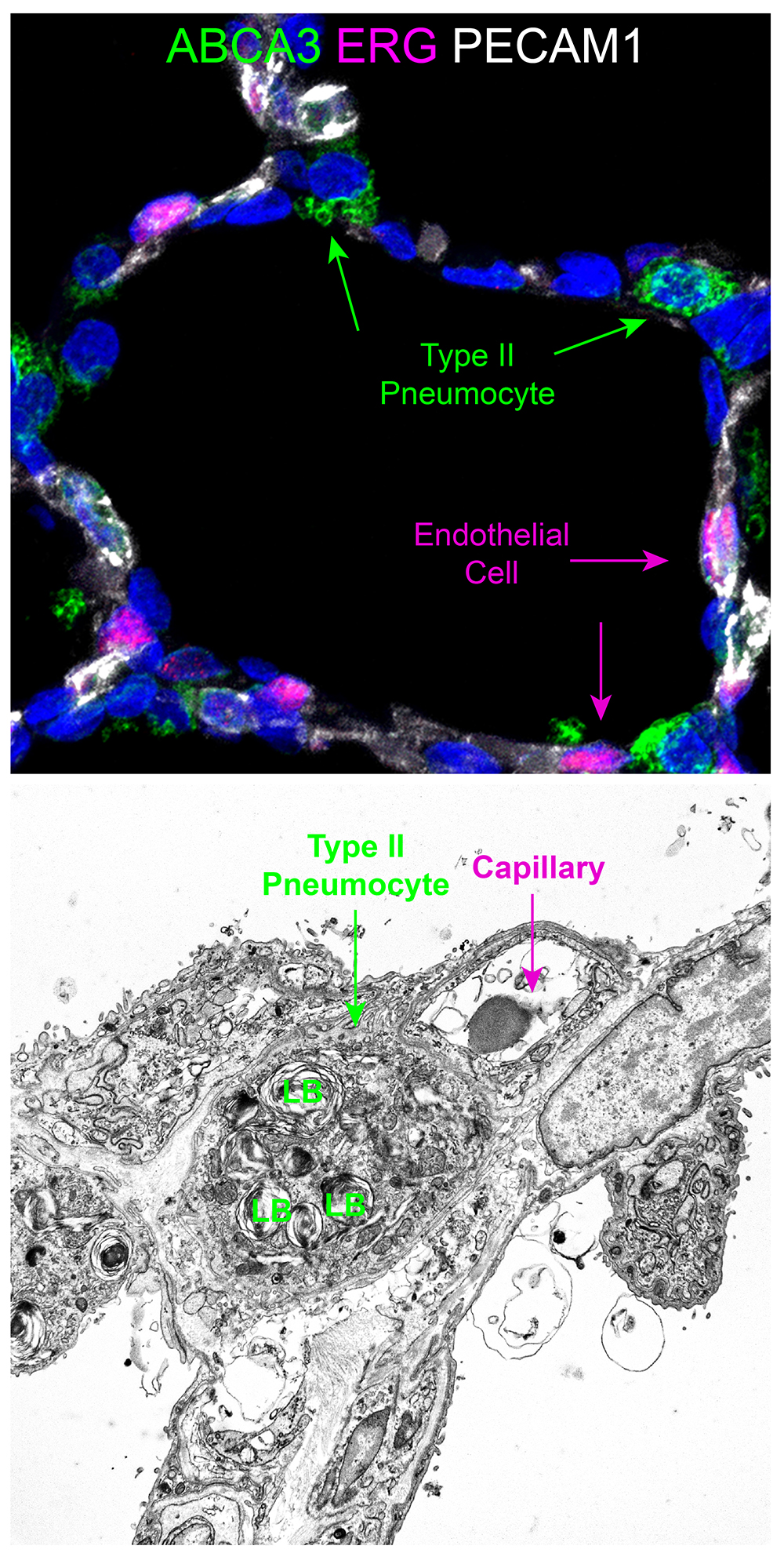
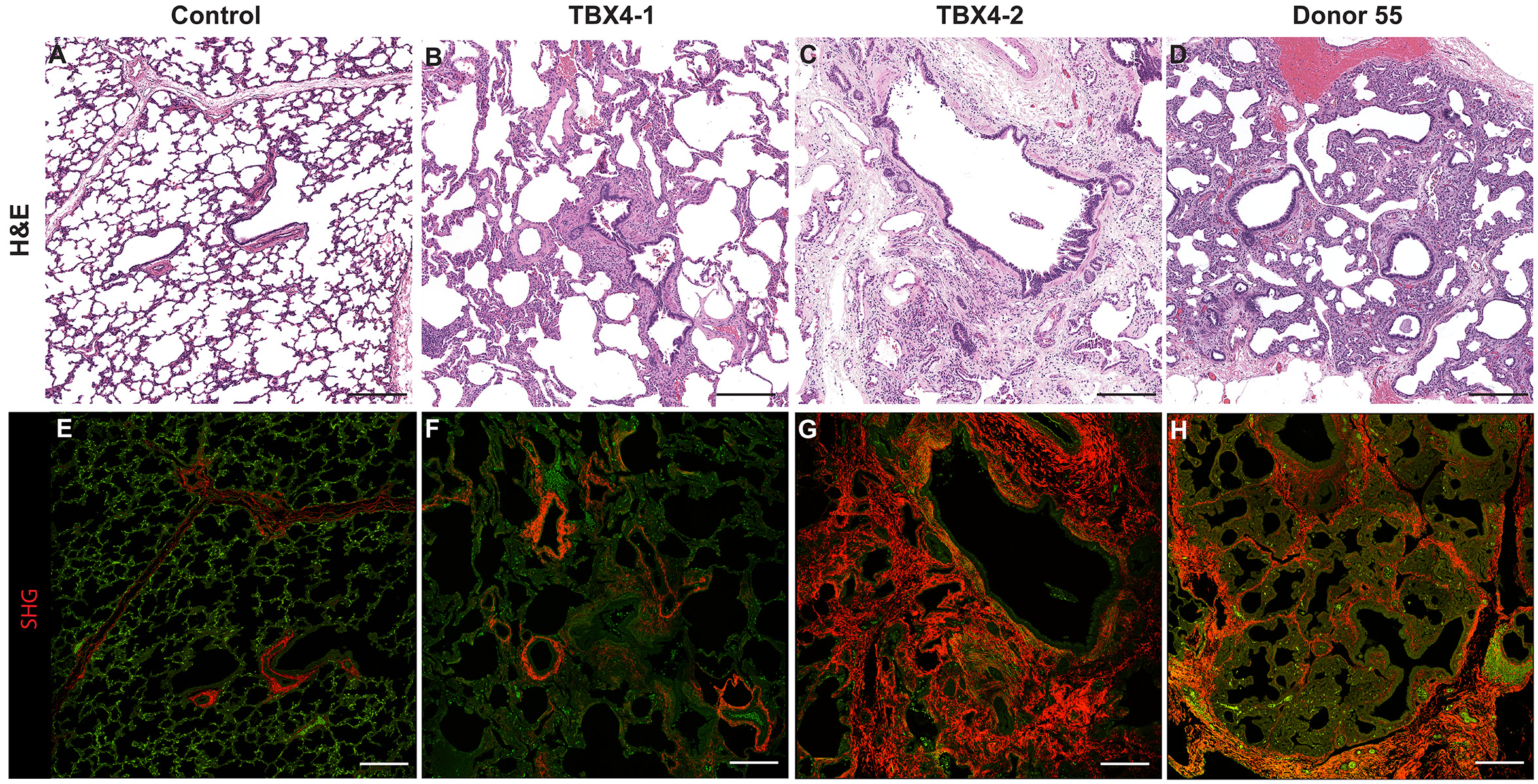
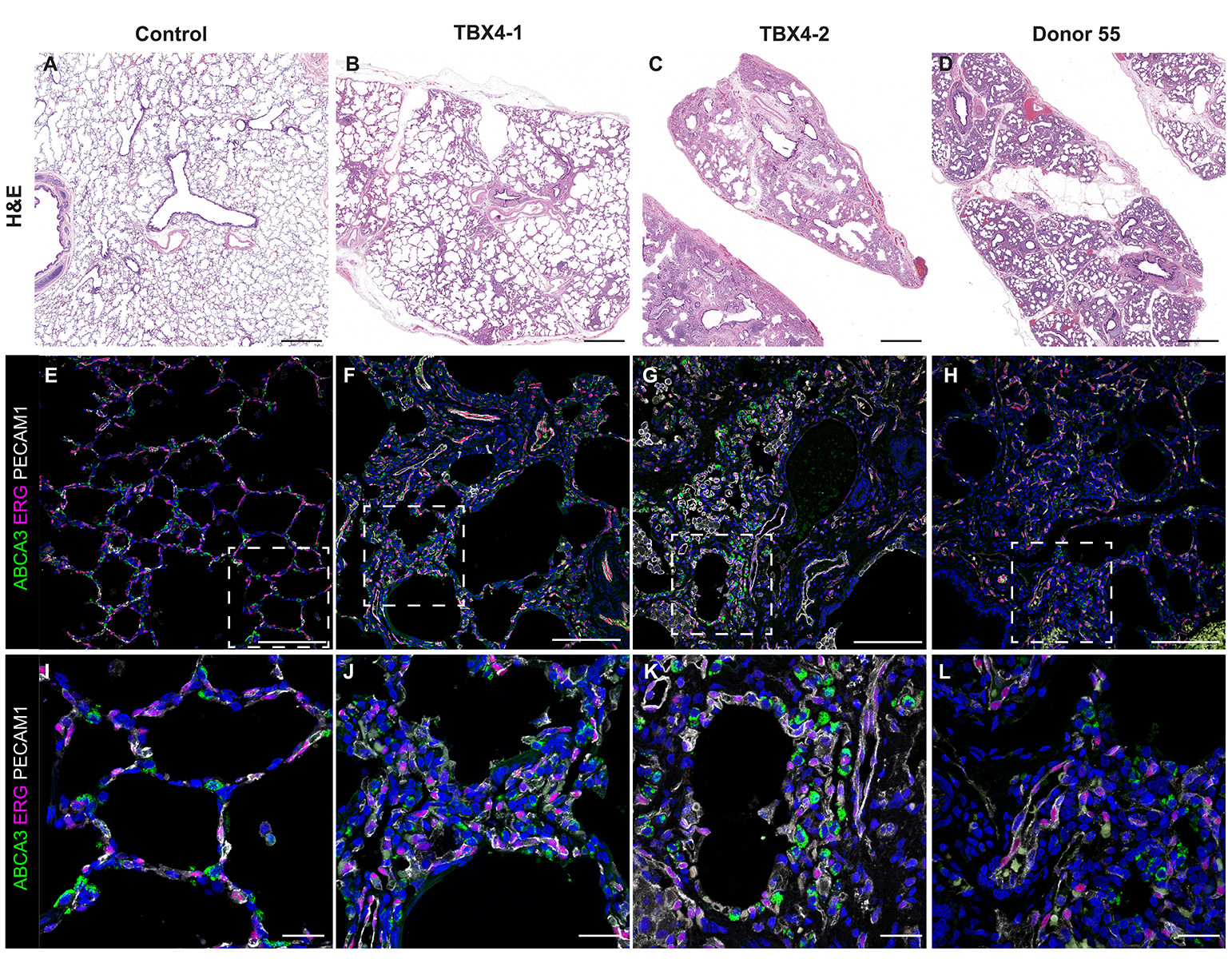
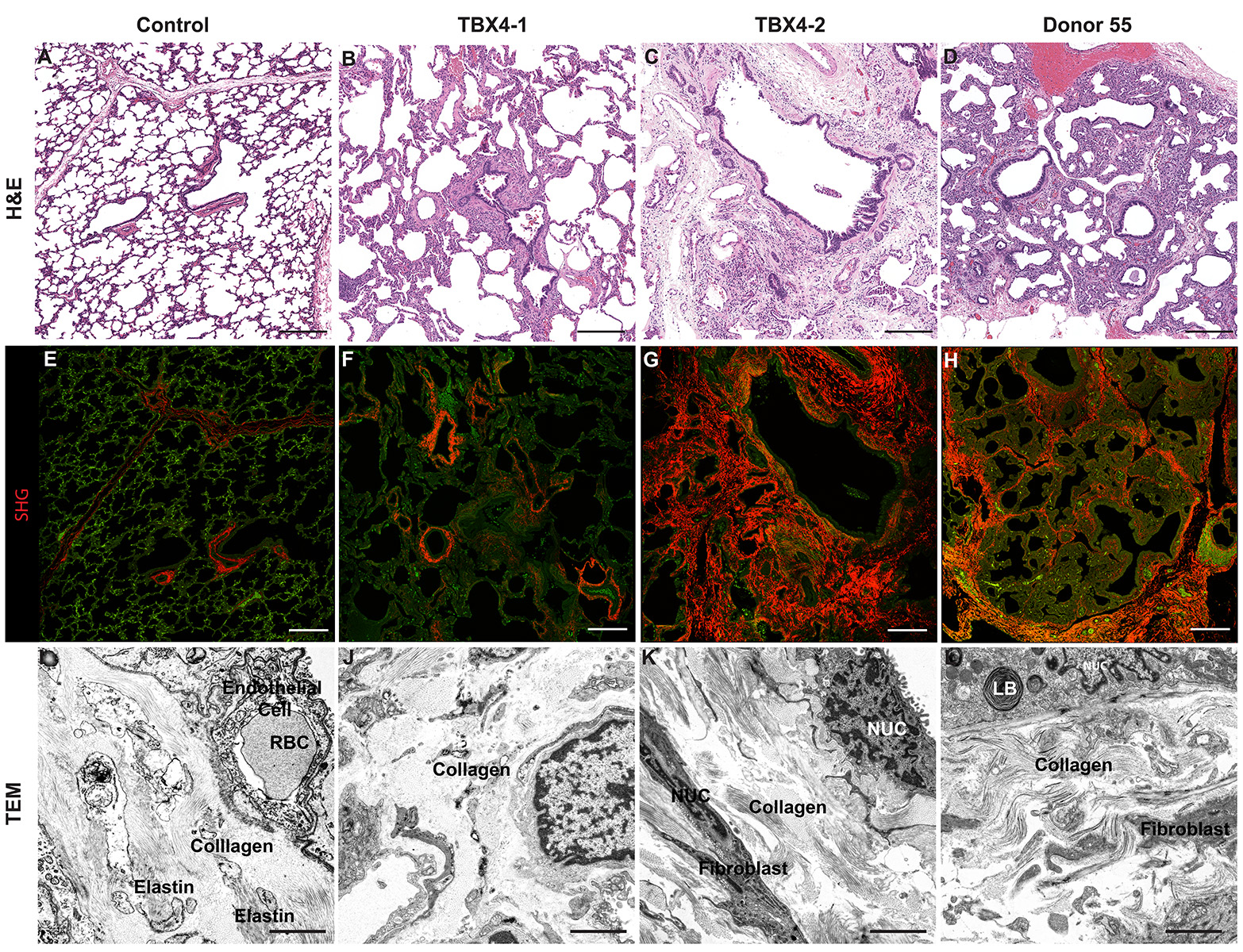
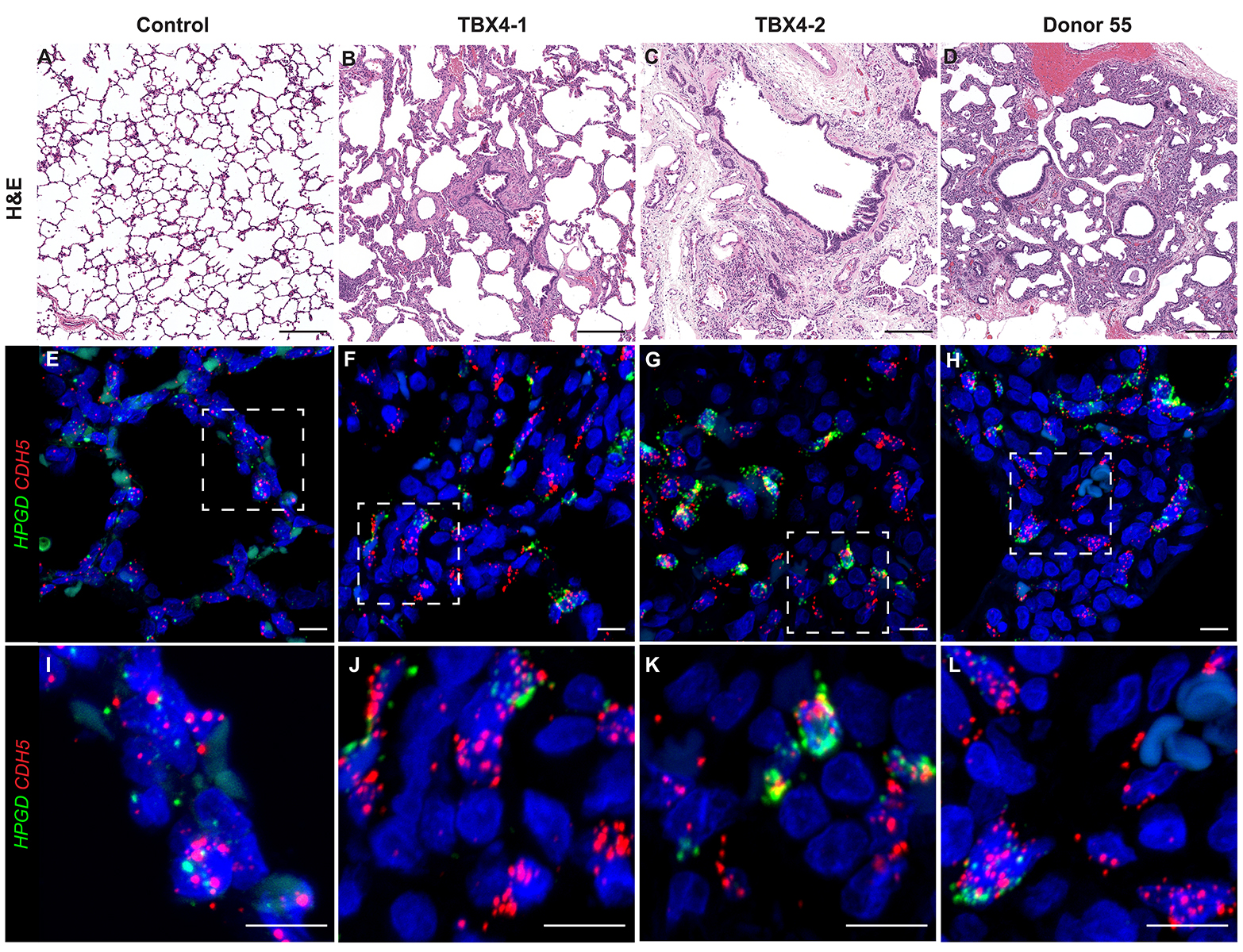
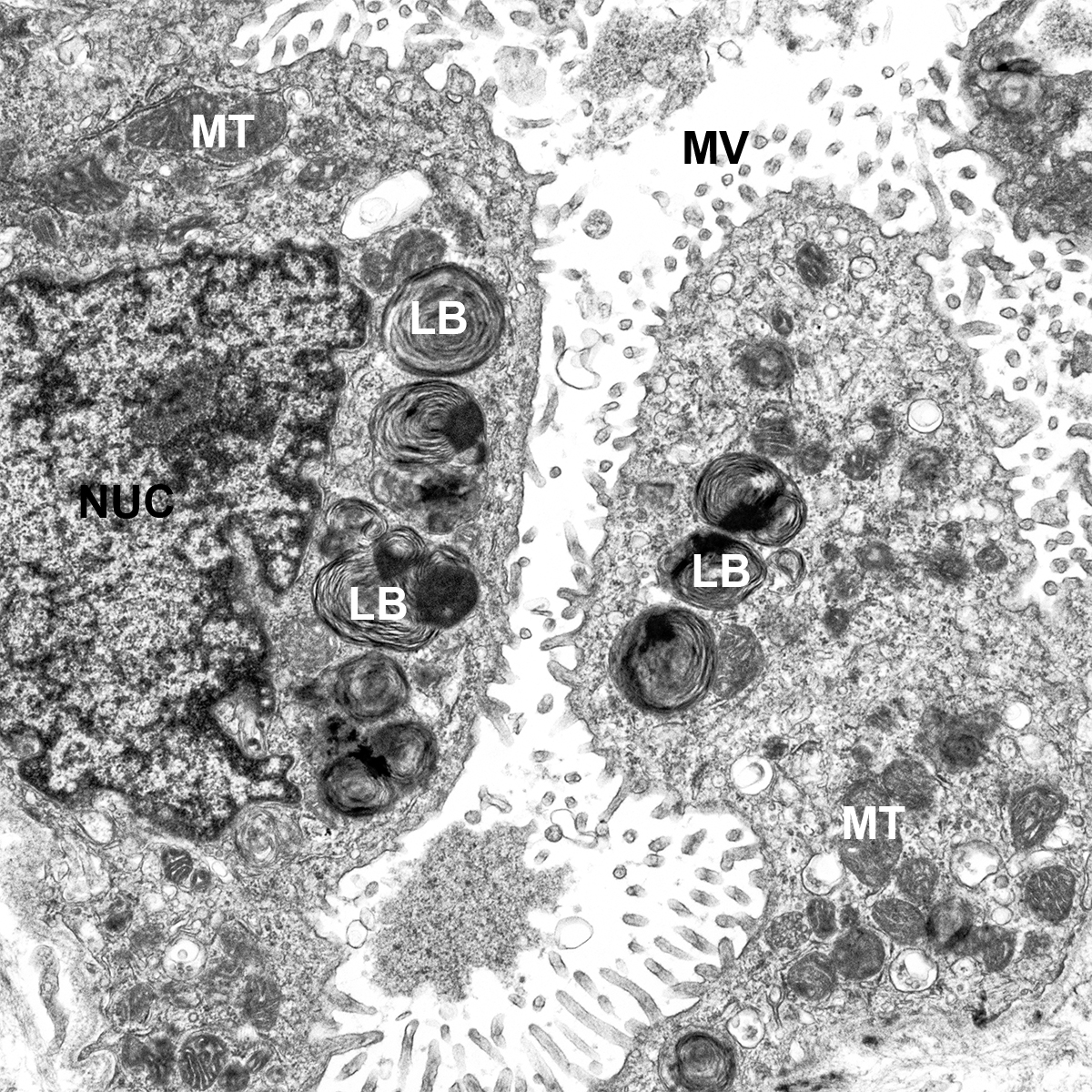
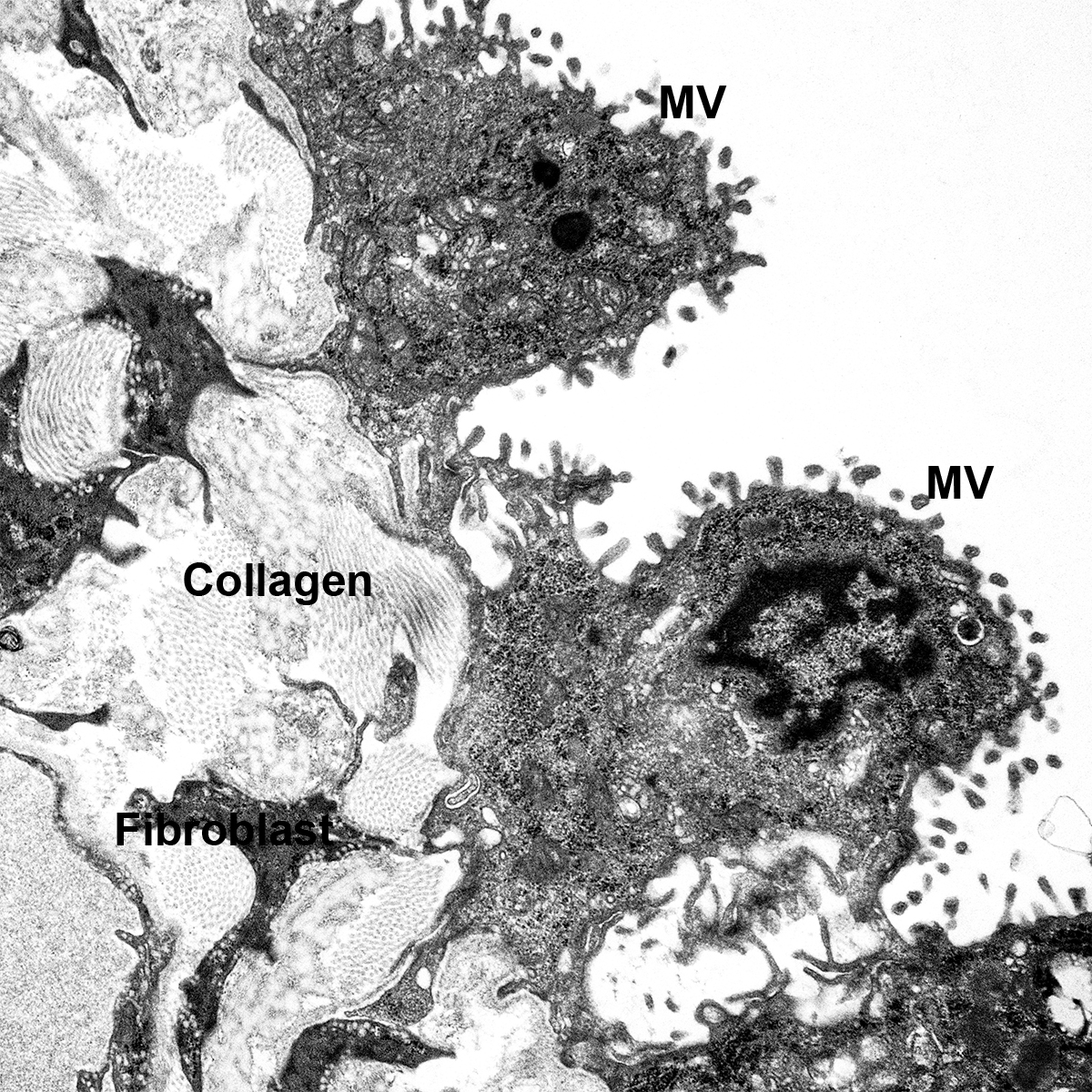
- Look at tissue under a light microscope and make notes of the various structures (i.e. cartilaginous airway, bronchioles, pulmonary arteries, veins, alveoli, etc.) present for each individual slide. This will help determine the appropriate antibodies to use to stain the various structures.
- Store at -20oC for short term storage or -80oC for long term storage for frozen sections. Paraffin sections can be stored at 4o
- Wash frozen tissue 2X in 1X PBS for 5 minutes each wash to rehydrate and remove OCT from tissue.
- Incubate slides in 4% PFA (to refix tissue) in 1X PBS, pH7.4 on tissue for 5 minutes.
- Wash 1X with 1X PBS.
- Paraffin sections are placed at 60oC for 2 hours to overnight to melt paraffin. Paraffin sections are then placed in xylene 3X for 10 minutes each, followed by 3X in 100% ETOH for 3 minutes each, 95% ETOH for 3 minutes each, and 70% ETOH for 3 minutes each. Slides are then placed in 1X PBS for 5 minutes to completely rehydrate tissue.
- If antigen retrieval is necessary to unmask nuclear antigens, antigen retrieval, pH 6.0 (times will vary according to microwave) is performed
- Fill plastic coplin jars with Antigen Retrieval buffer (10 mM sodium citrate, pH 6.0). Place in a Tupperware container and fill the Tupperware container half way with dH2O Heat in a microwave according to the instructions on the microwave. We use a GE microwave for 6.5 minutes at 1100 watts, 6 minutes at 440 watts, and finally 6 minutes at 440 watts. *We usually do a series of three runs to equal the time/temp because of evaporation (refill plastic coplin jars with dH2O).
- Cool on countertop, 15 min.
- Rinse with dH2O
- Wash1X PBS, pH 7.4 for 5 minutes.
- Block in 4% Goat serum/PBS-T, 2 hours at RT.
- Block for 1-2 hours at room temperature in 4% Serum/PBS/0.2% Triton X-100 with gentle shaking. Typically tissue will be blocked with goat, donkey, or horse serum. The serum species for blocking usually corresponds with the species the secondary antibodies are raised. For example with goat anti-rabbit secondary use goat serum for blocking.
- Remove blocking solution and add primary antibodies at pre-determined antibody titers diluted in the appropriate blocking solution. Antibodies are diluted in blocking solution and spun down in a µfuge for 10 minutes at full speed. Apply antibody solution to a cover well incubator (Electron Microscopy Sciences, Catalogue # 70324-20) and invert slide containing tissue onto antibody solution to overlay. Place slides containing antibodies in a humidity chamber and incubate overnight at 4oC.
- Wash 3-5X with 1X PBS + 0.2% triton X-100 for 5 minutes with gentle shaking at room temperature.
- The appropriate fluorescent secondary antibodies are diluted in blocking solution and spun down in a µfuge for 10 minutes at full speed. Apply the antibodies to the tissue and ncubate in the humidity chamber at room temperature for one hour.
- Wash 3-5X with 1X PBS + 0.2% triton X-100 for 5 minutes with gentle shaking at room temperature.
- Apply DAPI solution diluted at 1:2000 and incubate at room temp for 15 minutes.
- Wash 3-5X with 1X PBS + 0.2% triton X-100 for 5 minutes with gentle shaking at room temperature.
- Wash 3X with 1X PB without NaCl or triton X-100.
- Flick the slides to remove excess PB and add a drop of Prolong Gold anti-fade mounting medium (Invitrogen, P36930).
- Apply a Gold Seal Coverslip (#1.5, Cat# 63792-01 Electron Microscopy Sciences, 24 X 50 mm).
- Allow the Prolong Gold to cure overnight in a light sealed box at room temperature and then store the slides in a slide box at 4oC.
- Obtain 1024 X 1024 z-stacks of the slides on an inverted Confocal Microscope at various magnifications.
- Process the z-stacks in Imaris (Bitplane) imaging software. Export as .tiff files at 600 DPI.
- Levels adjustment in Adobe Photoshop can be used to increase the brightness of the image as needed.
Appendix
Antigen Retrieval Solution
18ml Solution A- 0.1M Citric Acid (Sigma, C1909) pH 2.5
82ml Reagent B- 0.1M Sodium Citrate (Sigma, S4641 ) pH 8.2
1L dH2O
pH 6.0