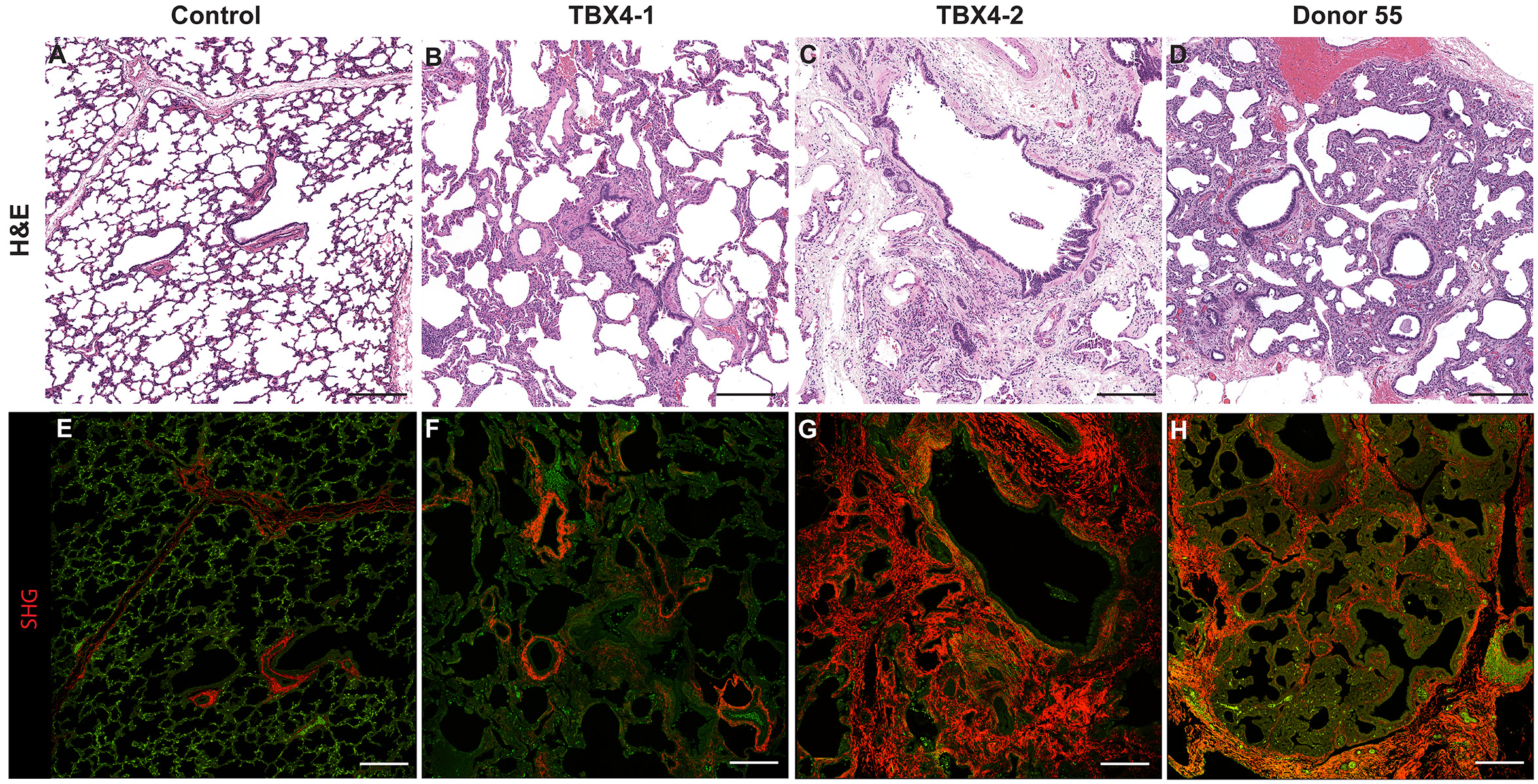
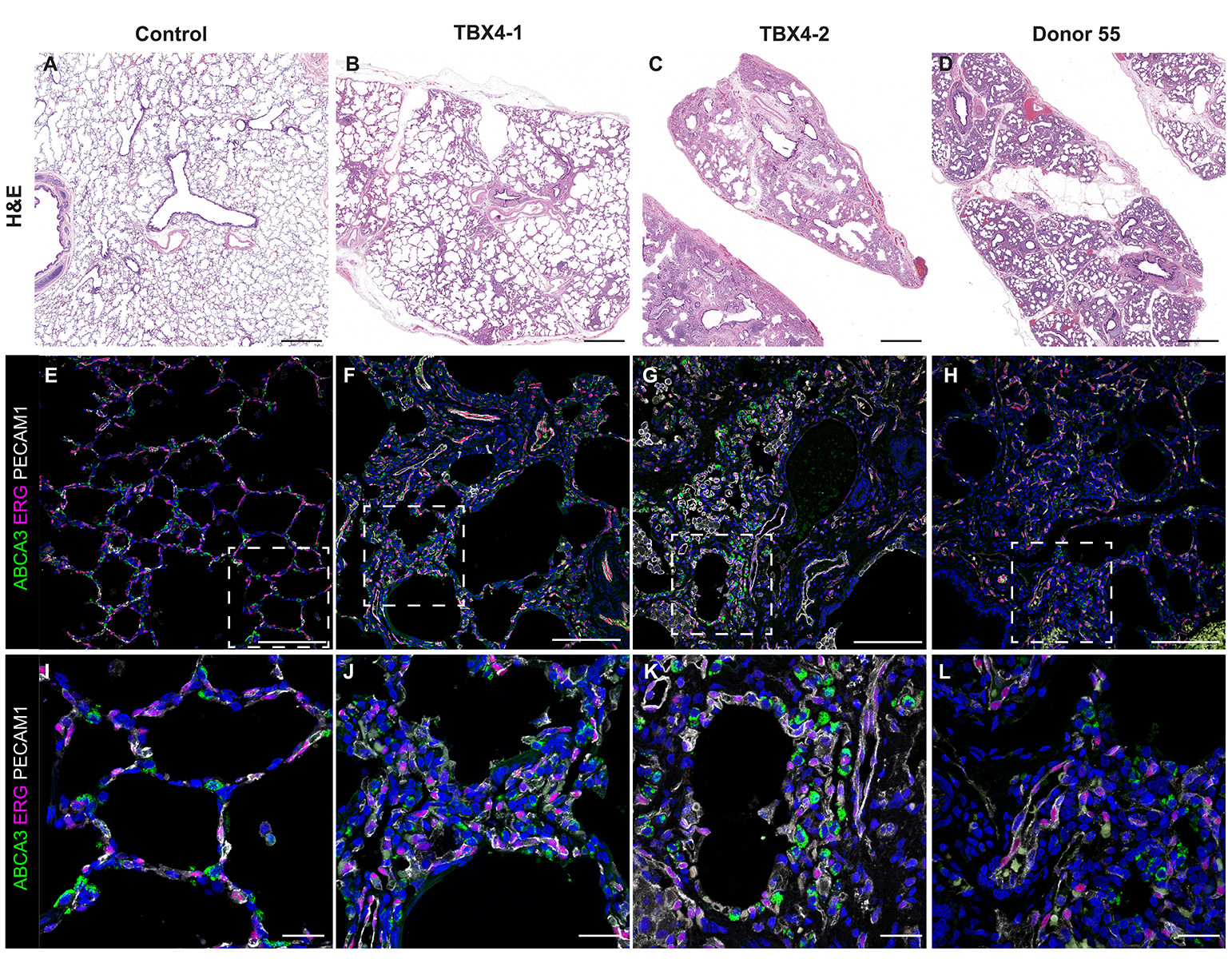
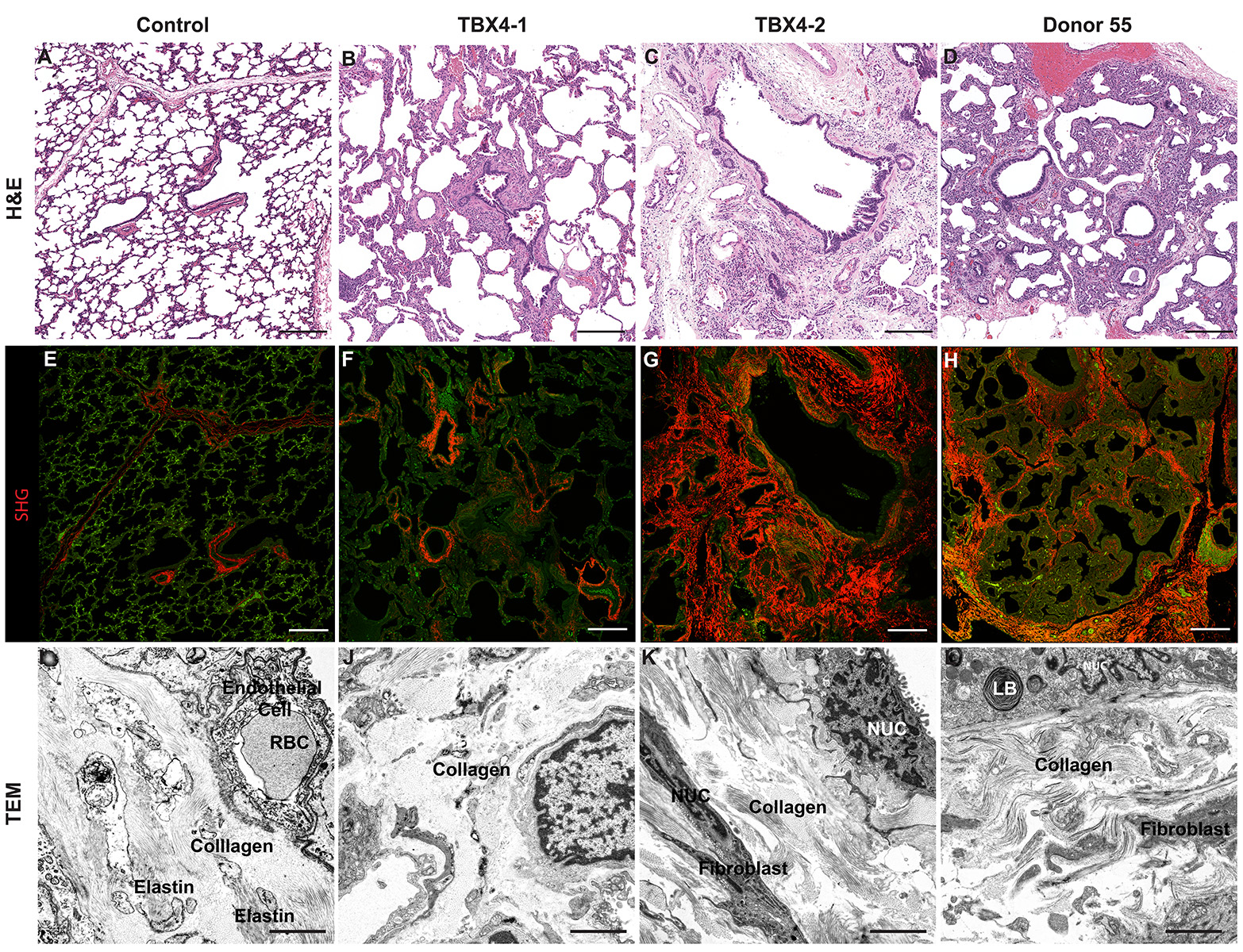
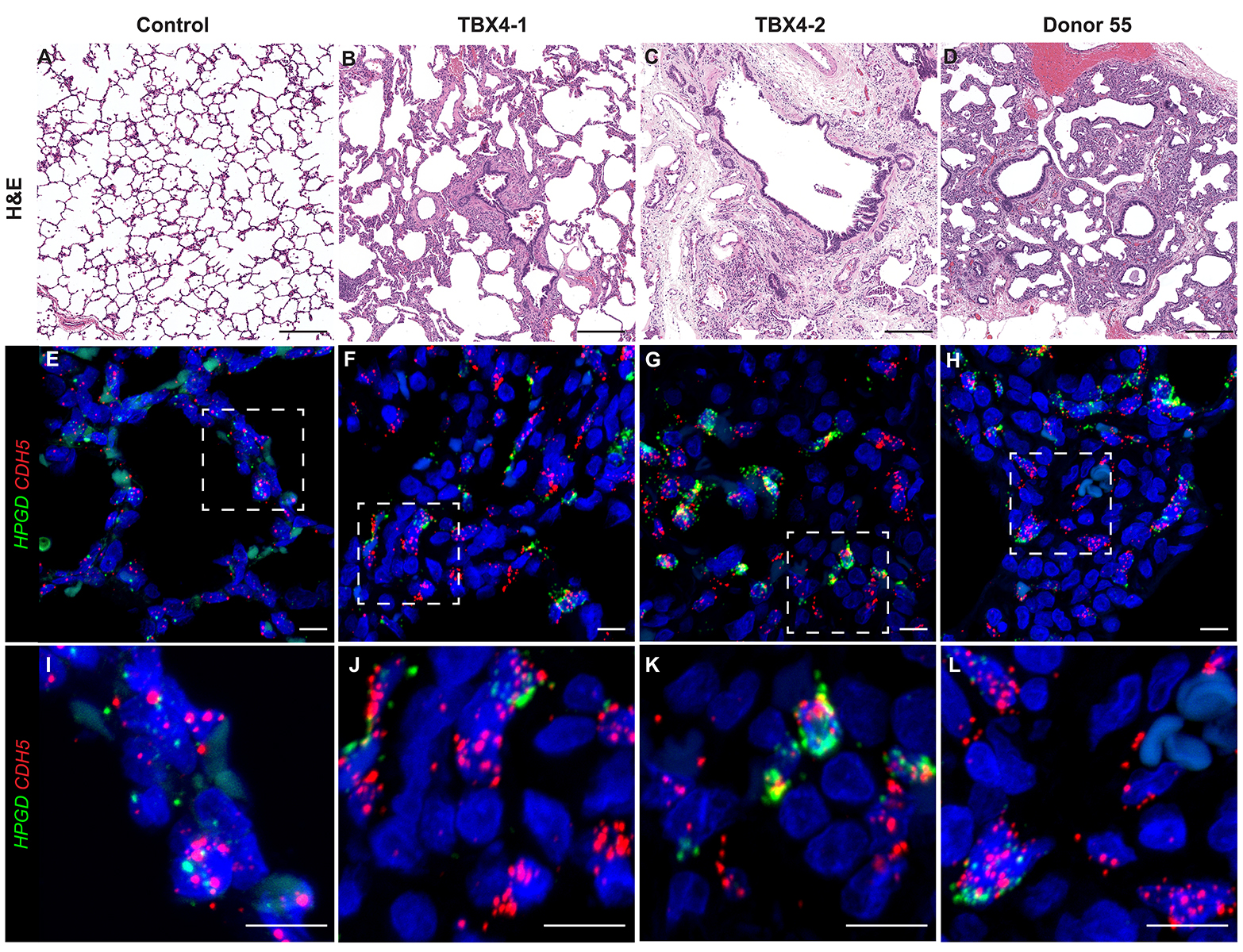
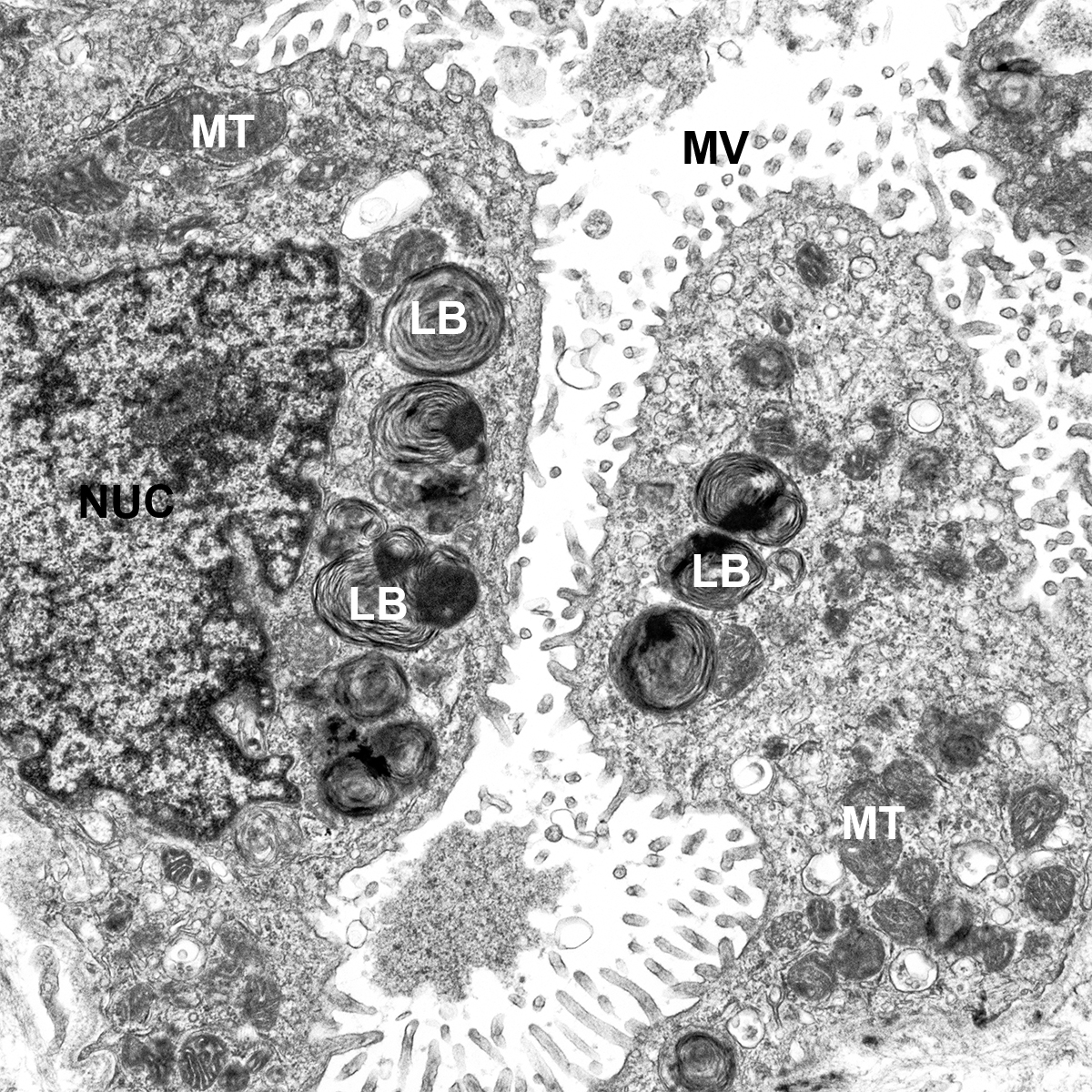
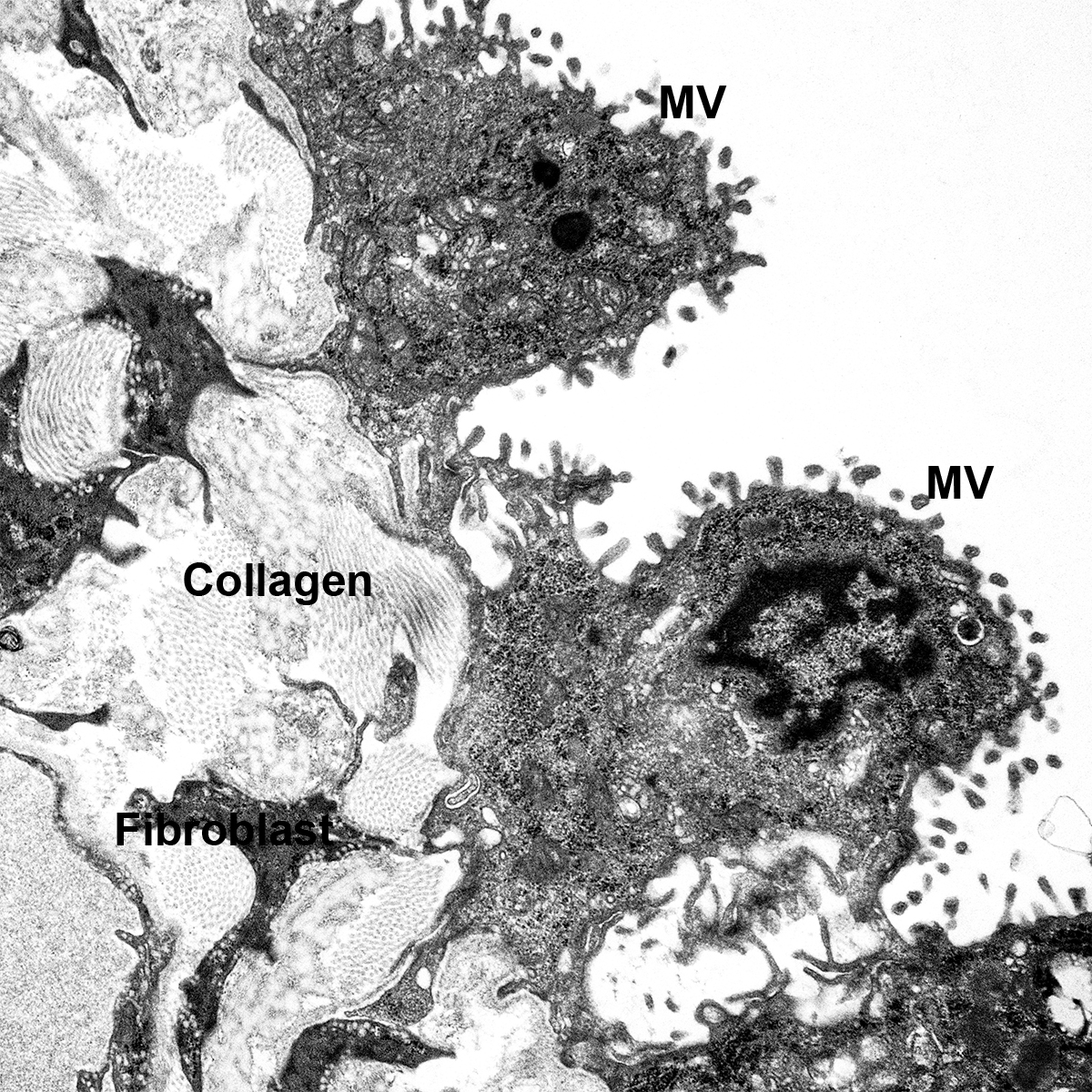
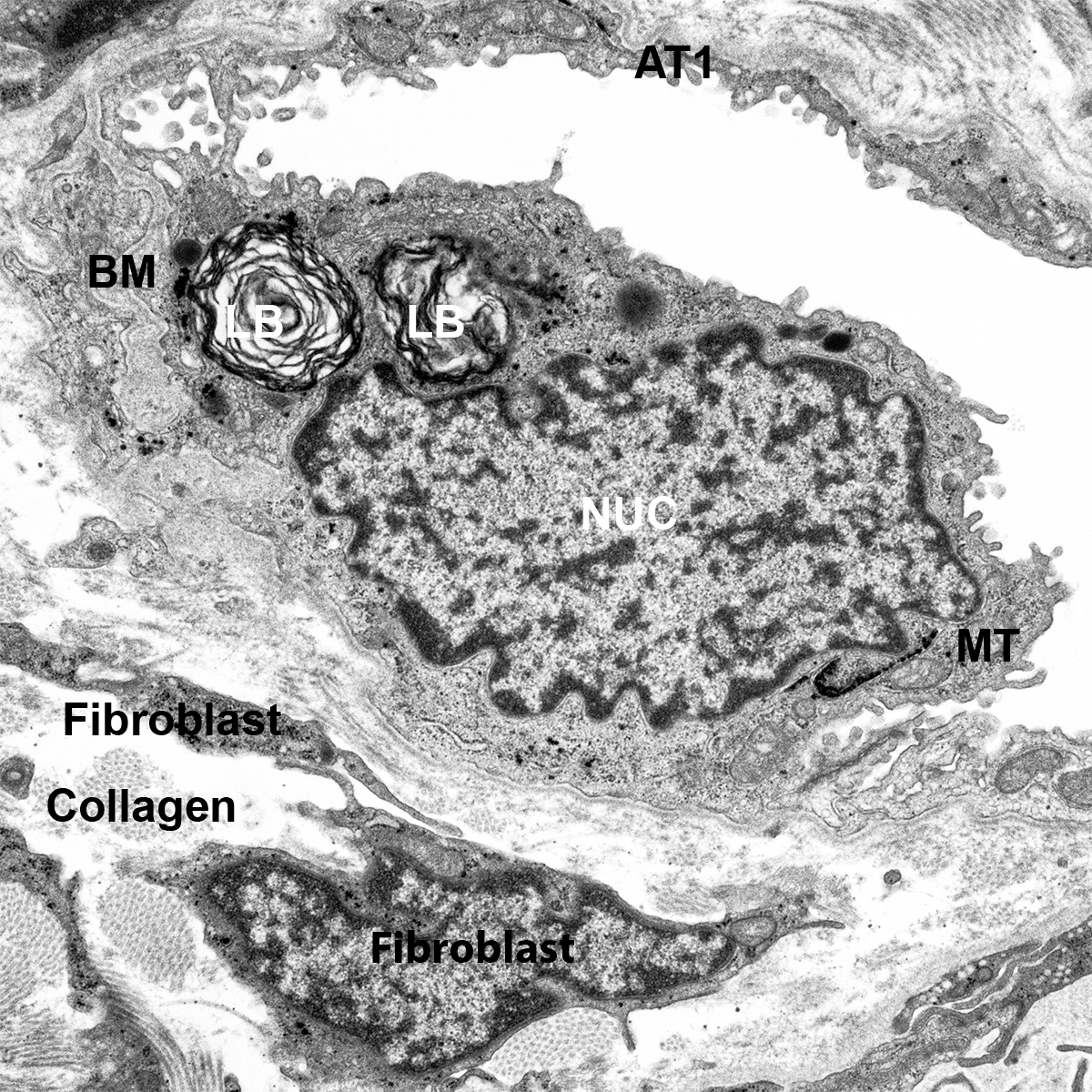
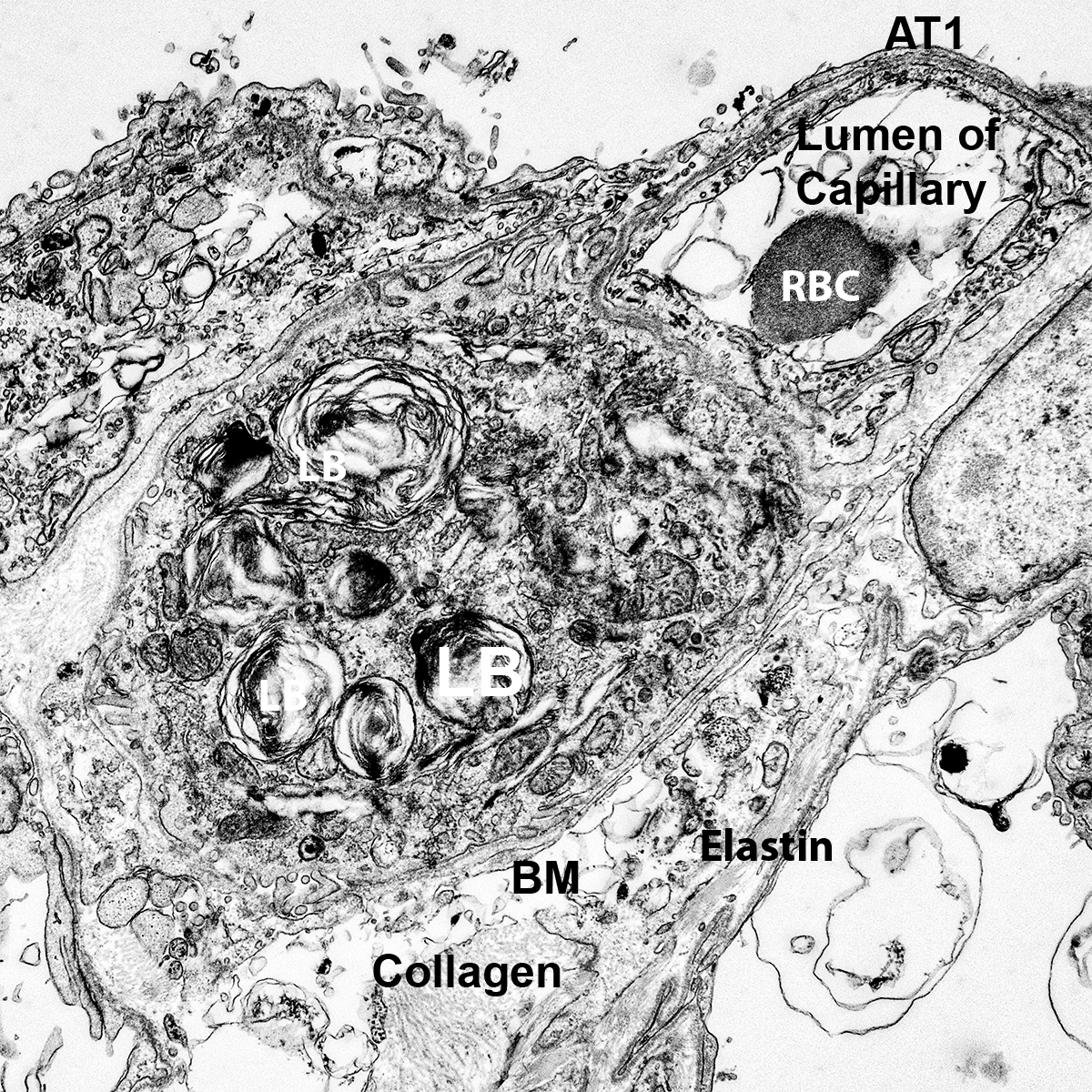
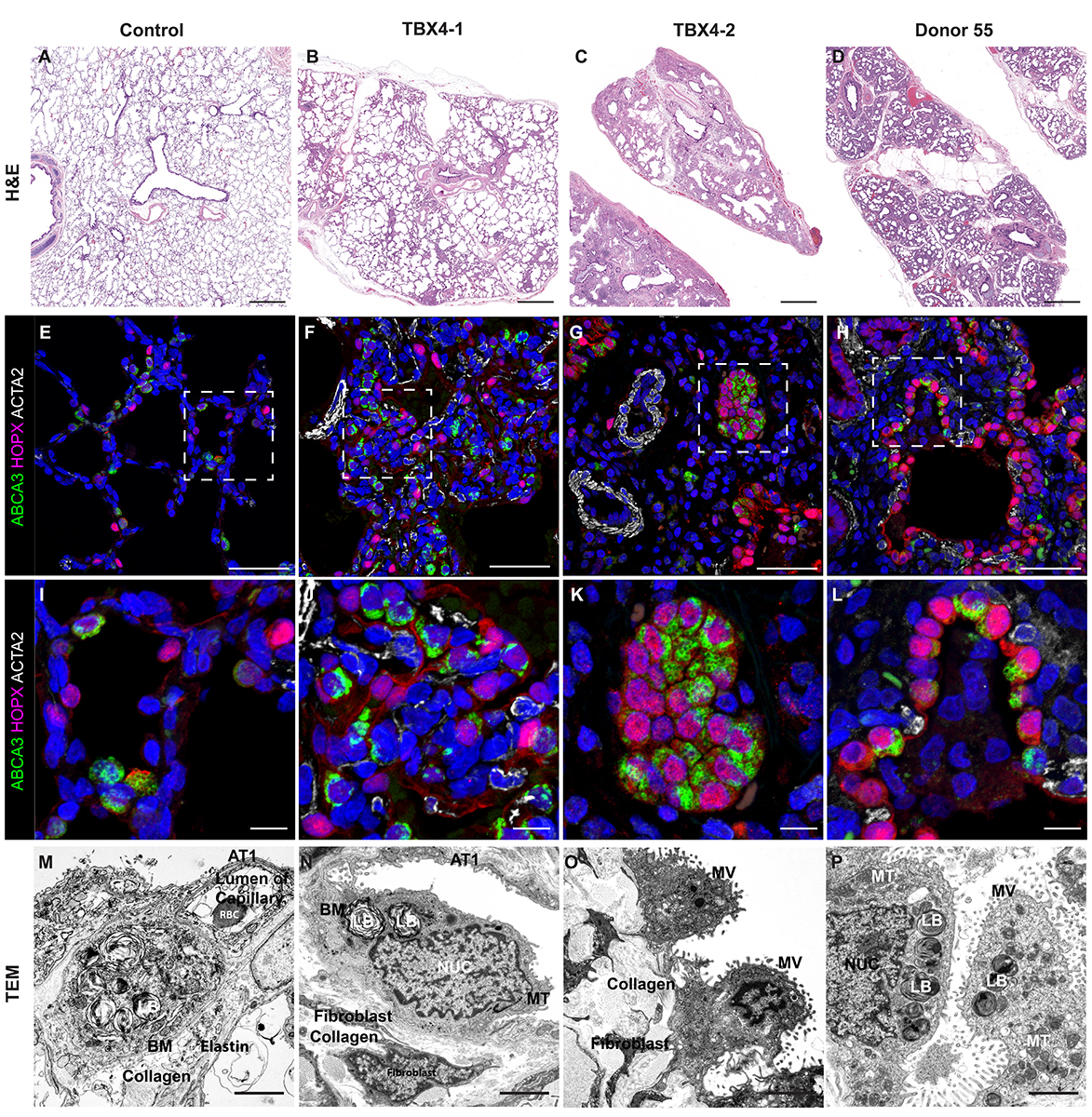
H&E Staining from PBR 3653 Patient with Cystic Fibrosis
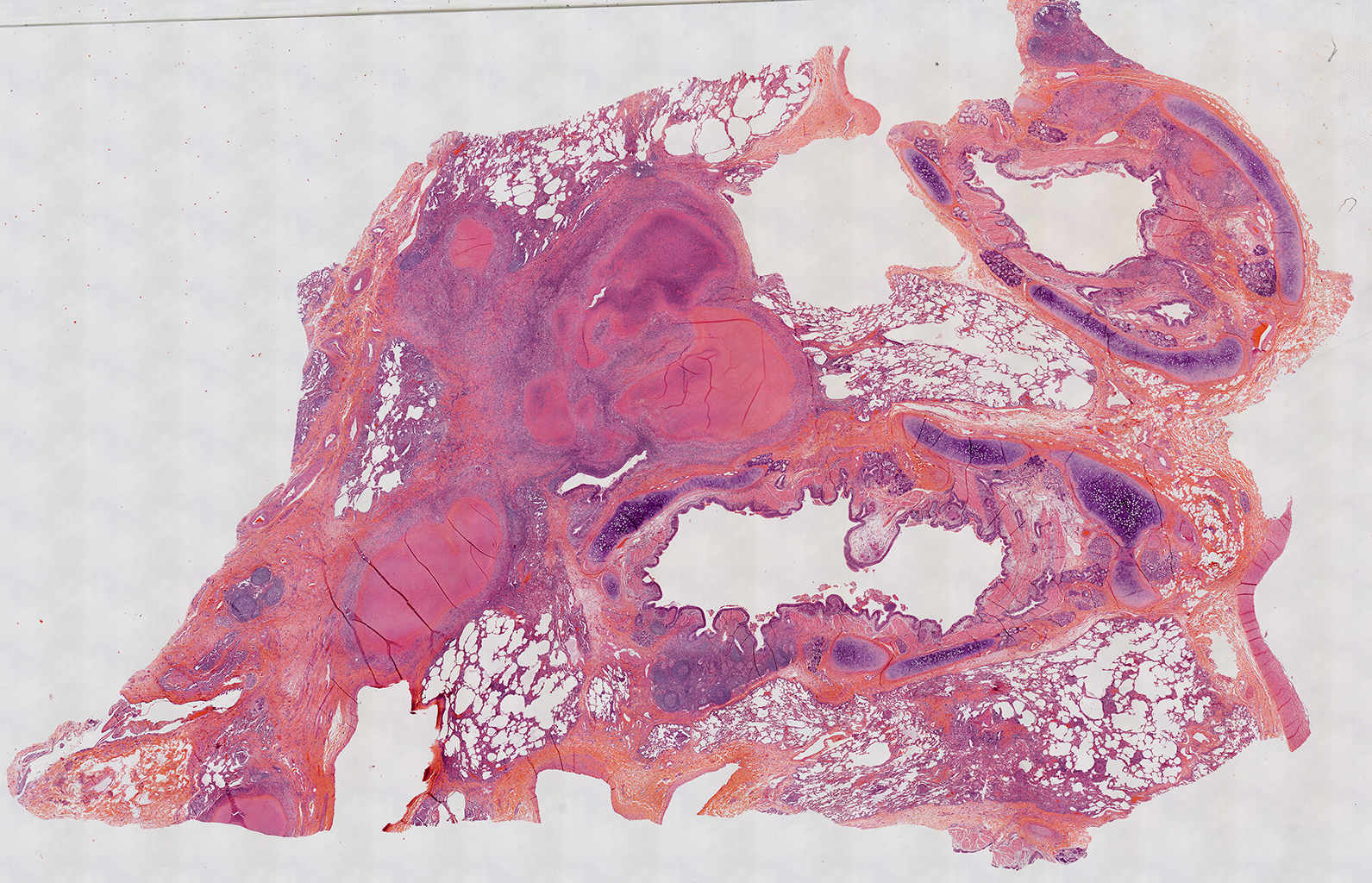
H&E Staining from PBR 3653 Patient with Cystic Fibrosis
4X Tilescan
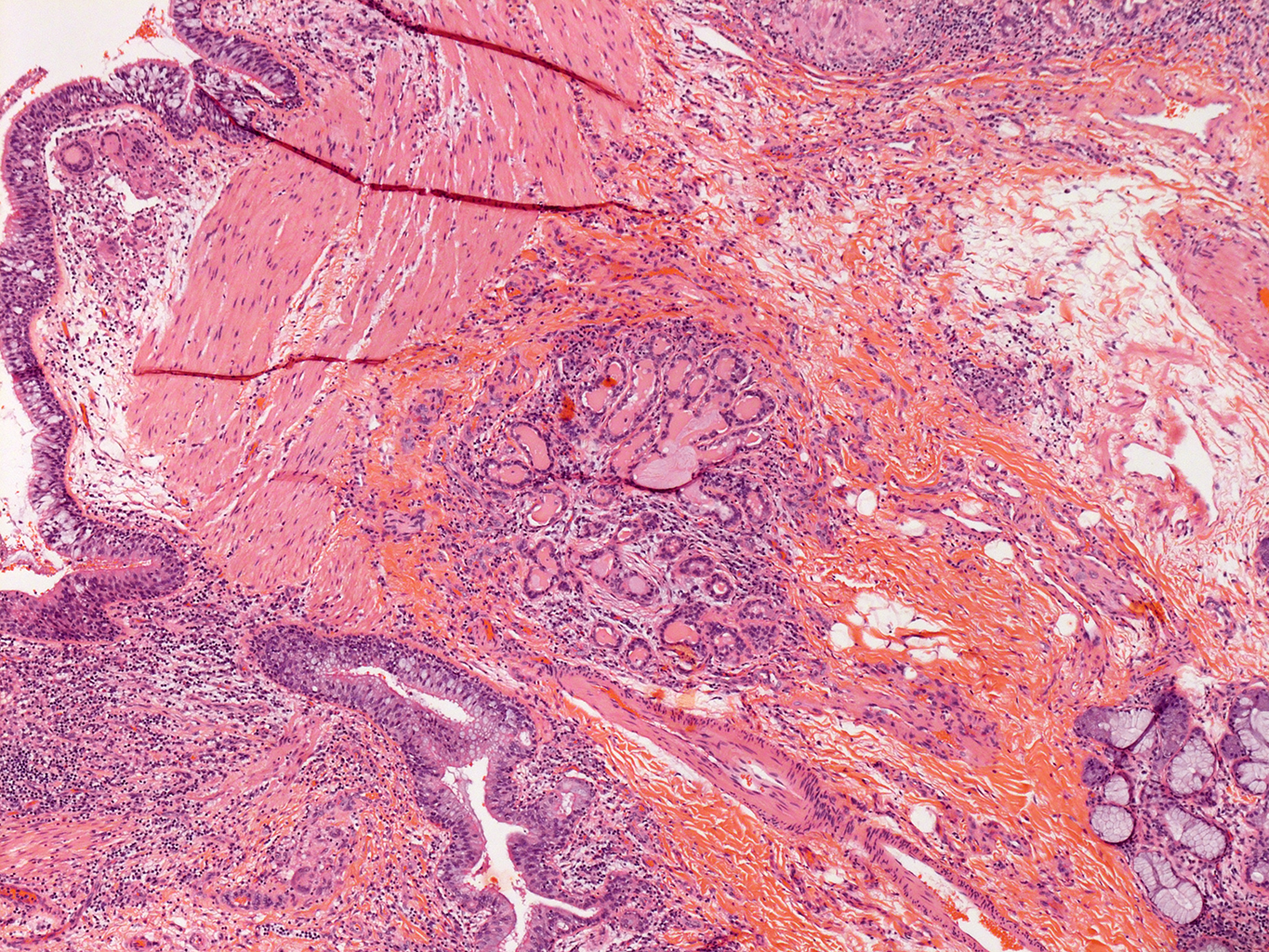
H&E Staining from PBR 3653 Patient with Cystic Fibrosis
4X
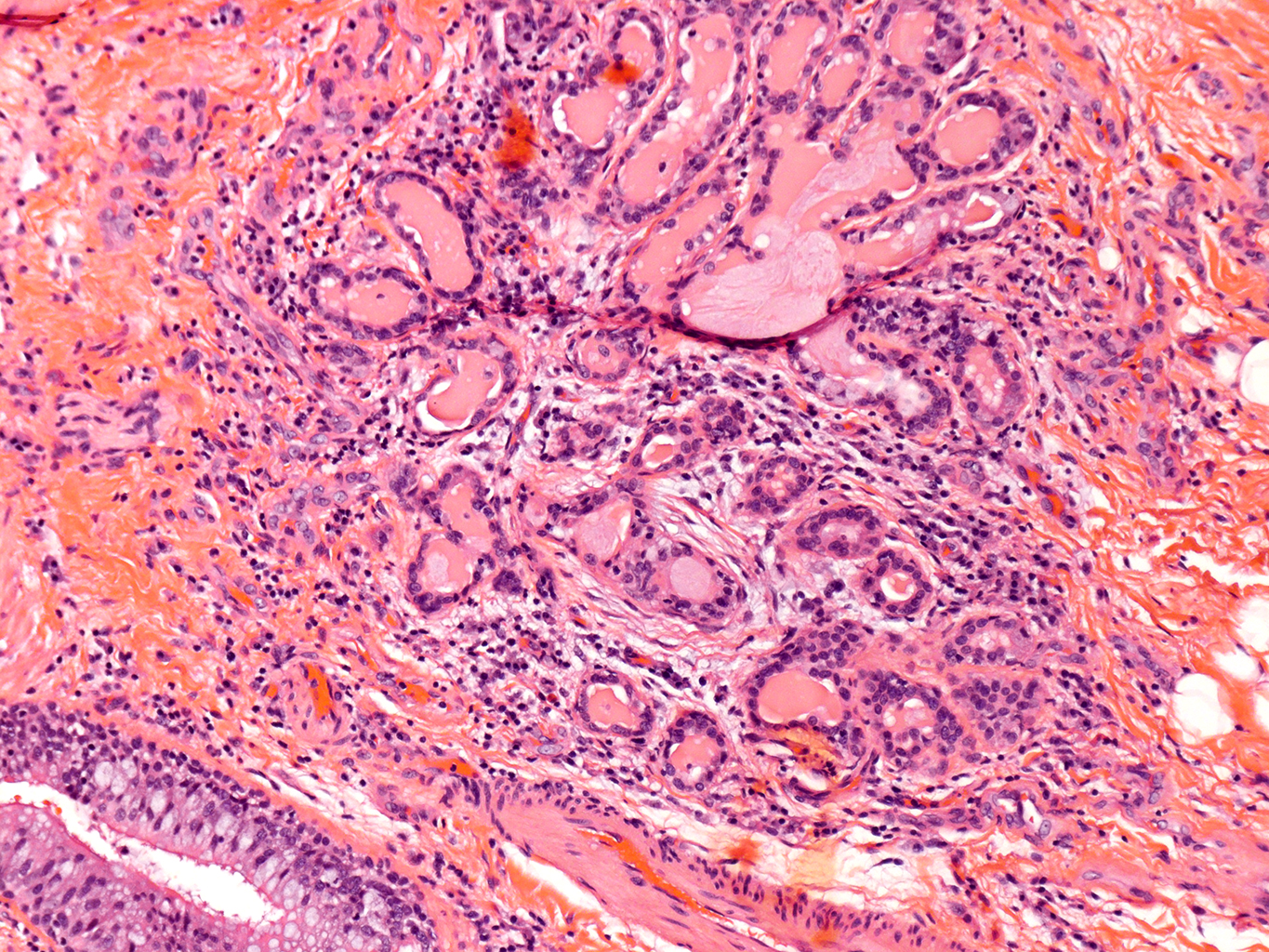
H&E Staining from PBR 3653 Patient with Cystic Fibrosis
10X
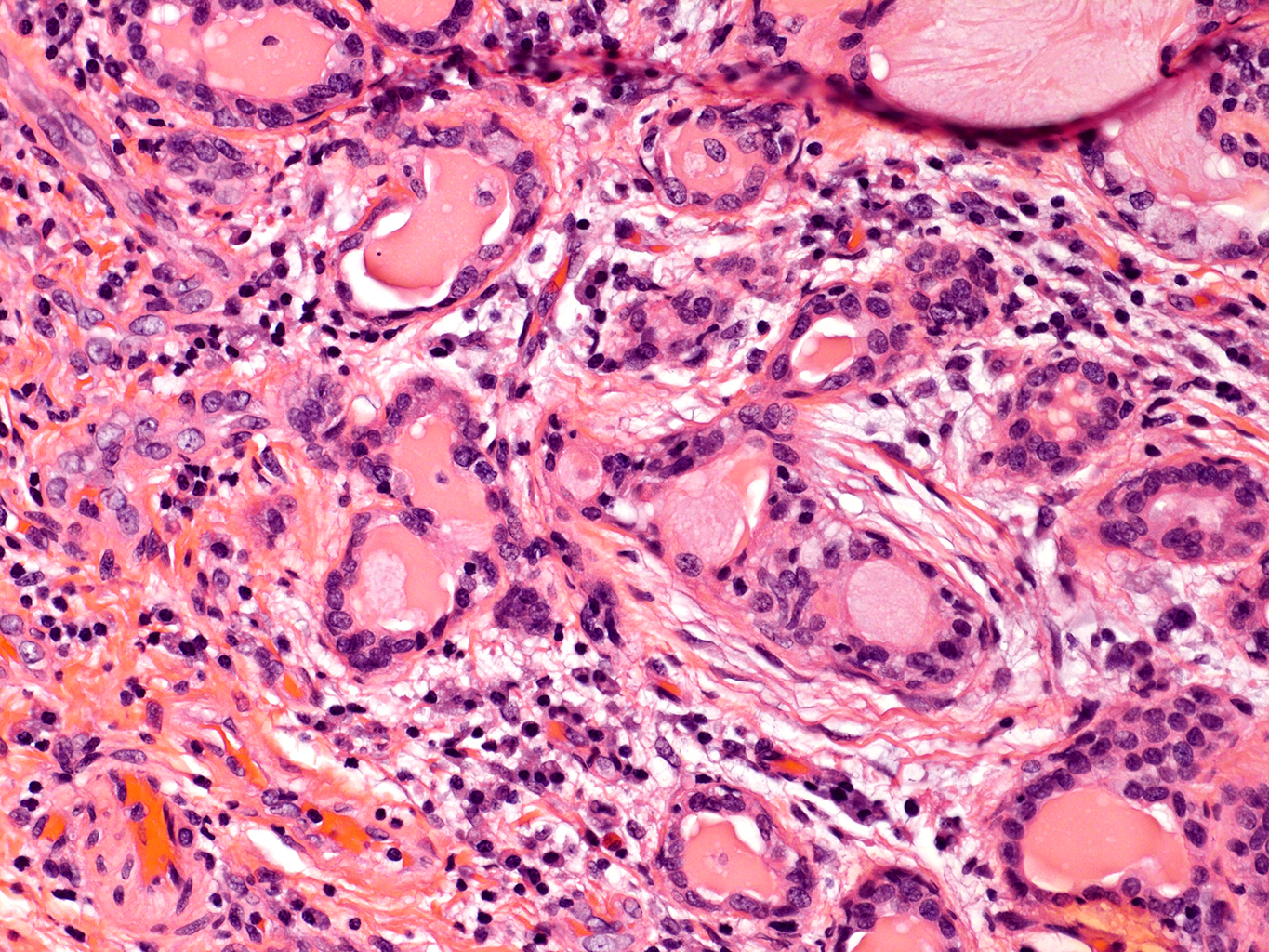
H&E Staining from PBR 3653 Patient with Cystic Fibrosis
20X
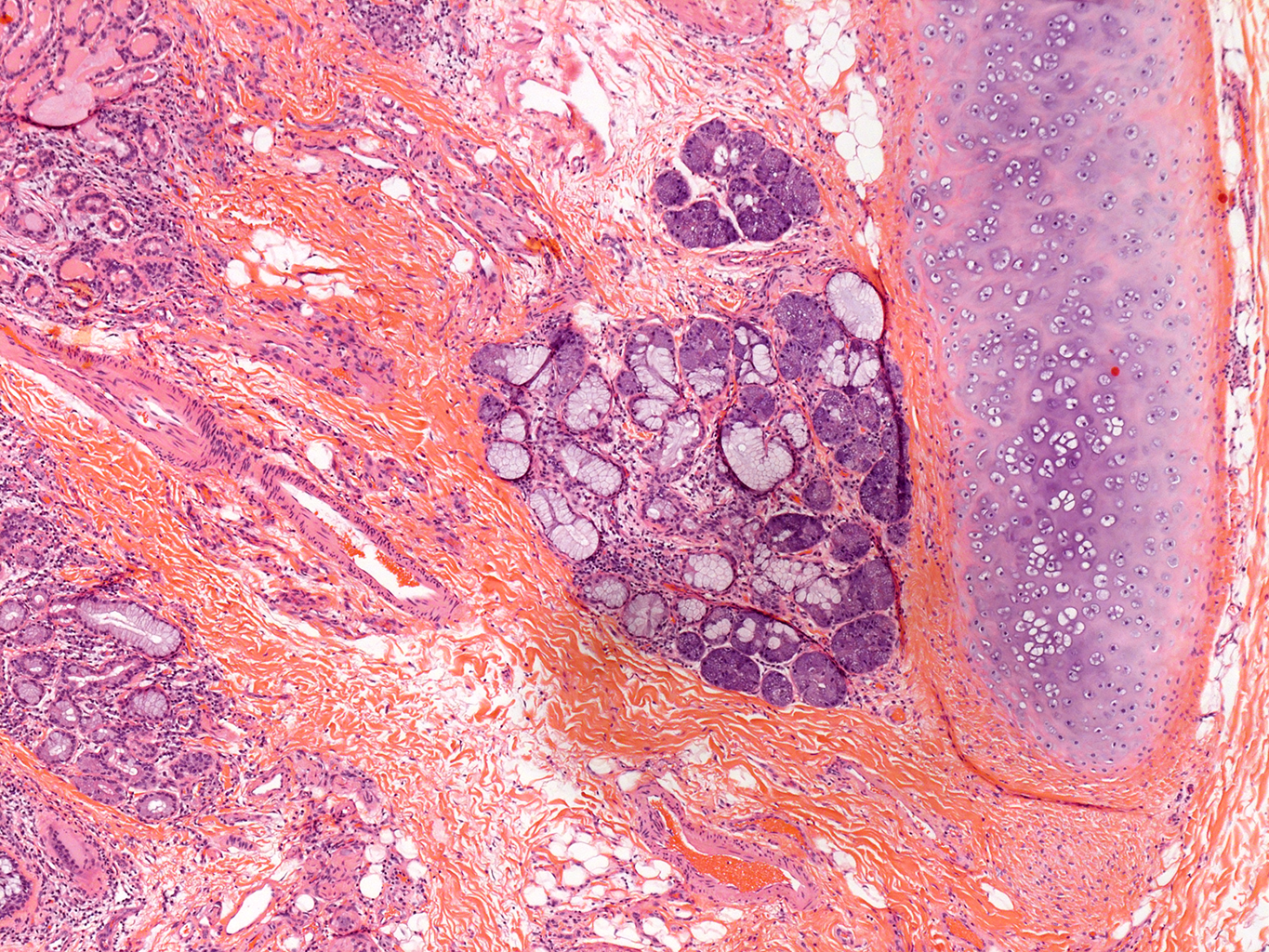
H&E Staining from PBR 3653 Patient with Cystic Fibrosis
4X
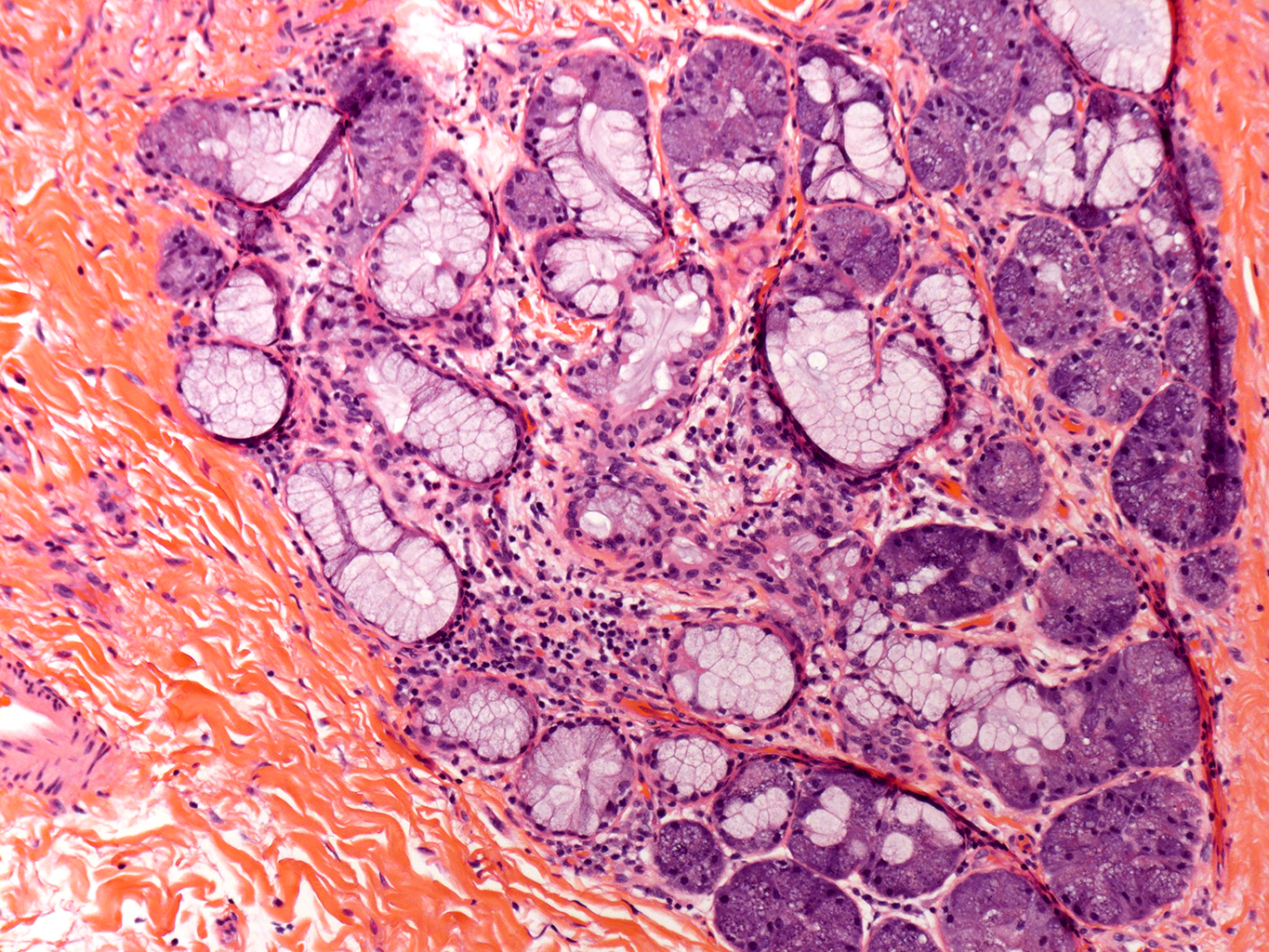
H&E Staining from PBR 3653 Patient with Cystic Fibrosis
10X
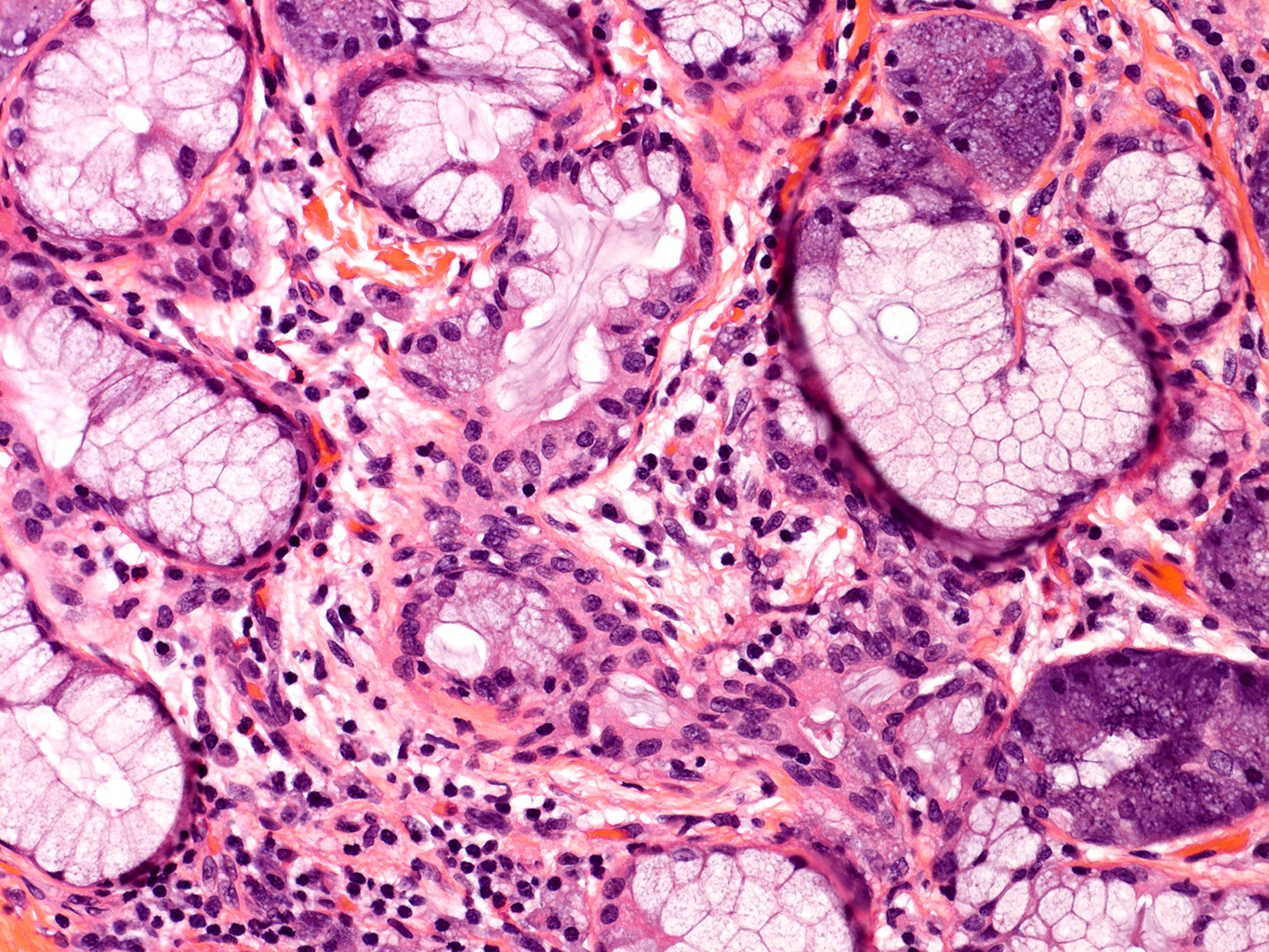
H&E Staining from PBR 3653 Patient with Cystic Fibrosis
20X
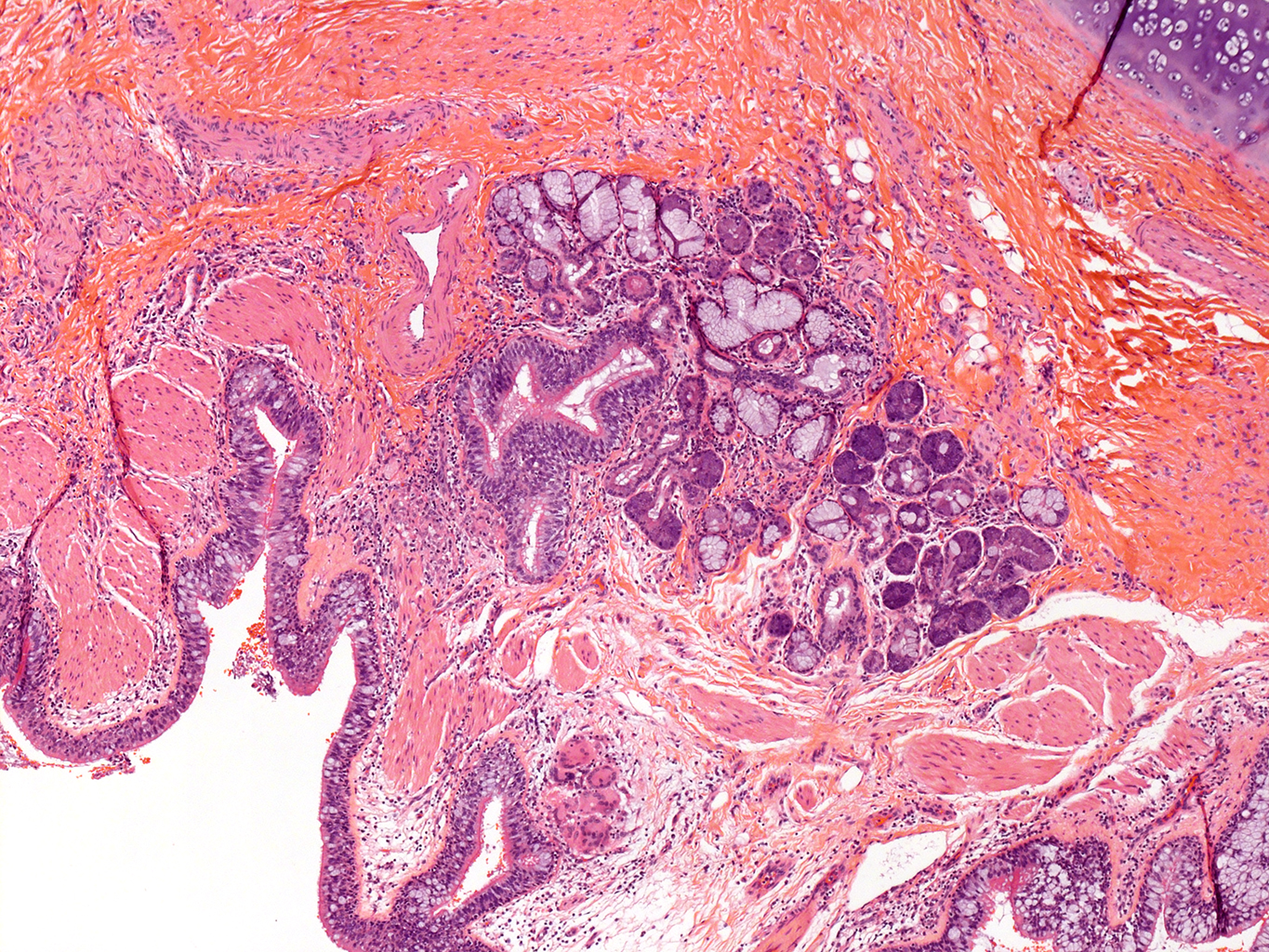
H&E Staining from PBR 3653 Patient with Cystic Fibrosis
4X
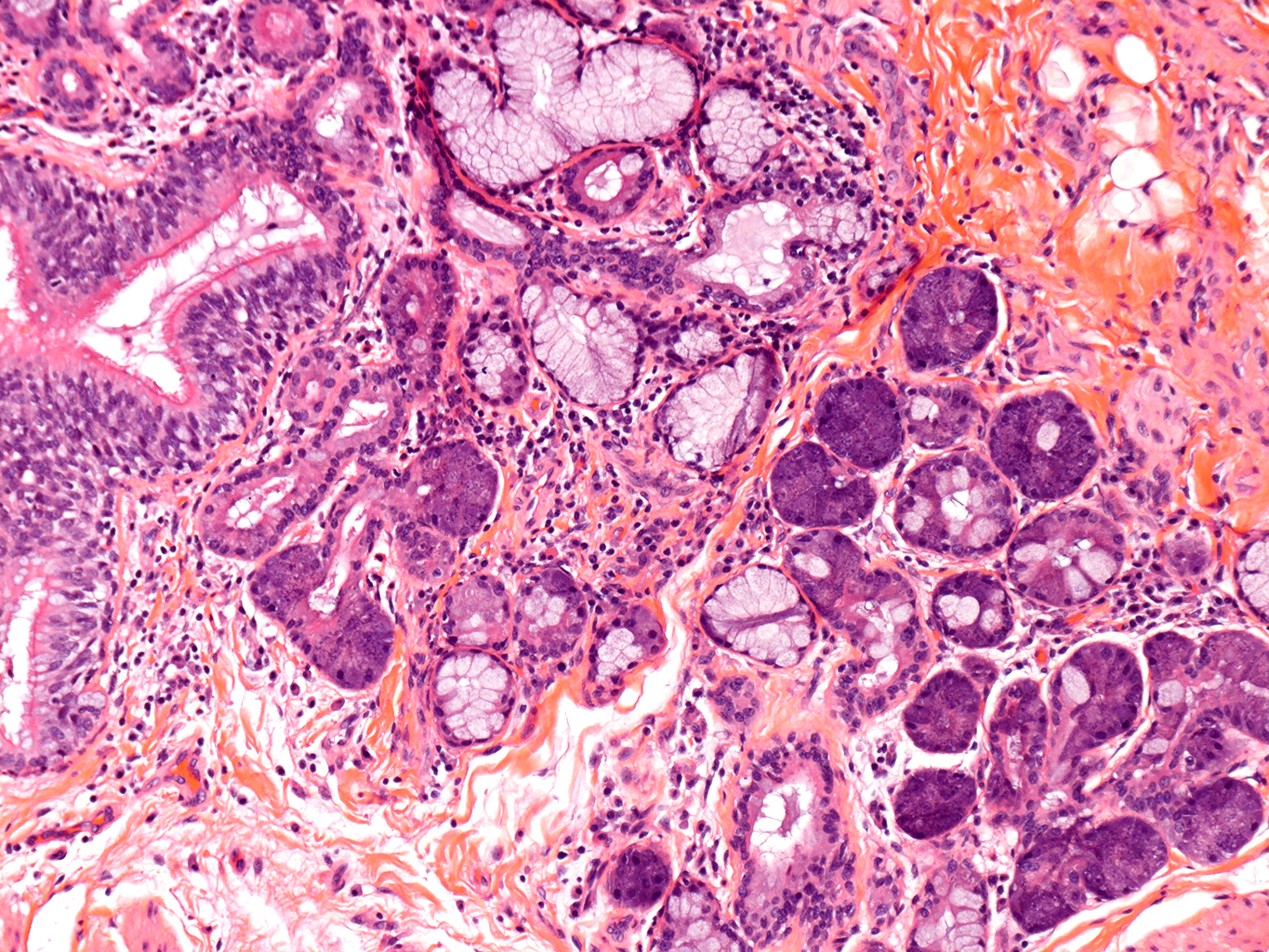
H&E Staining from PBR 3653 Patient with Cystic Fibrosis
10X
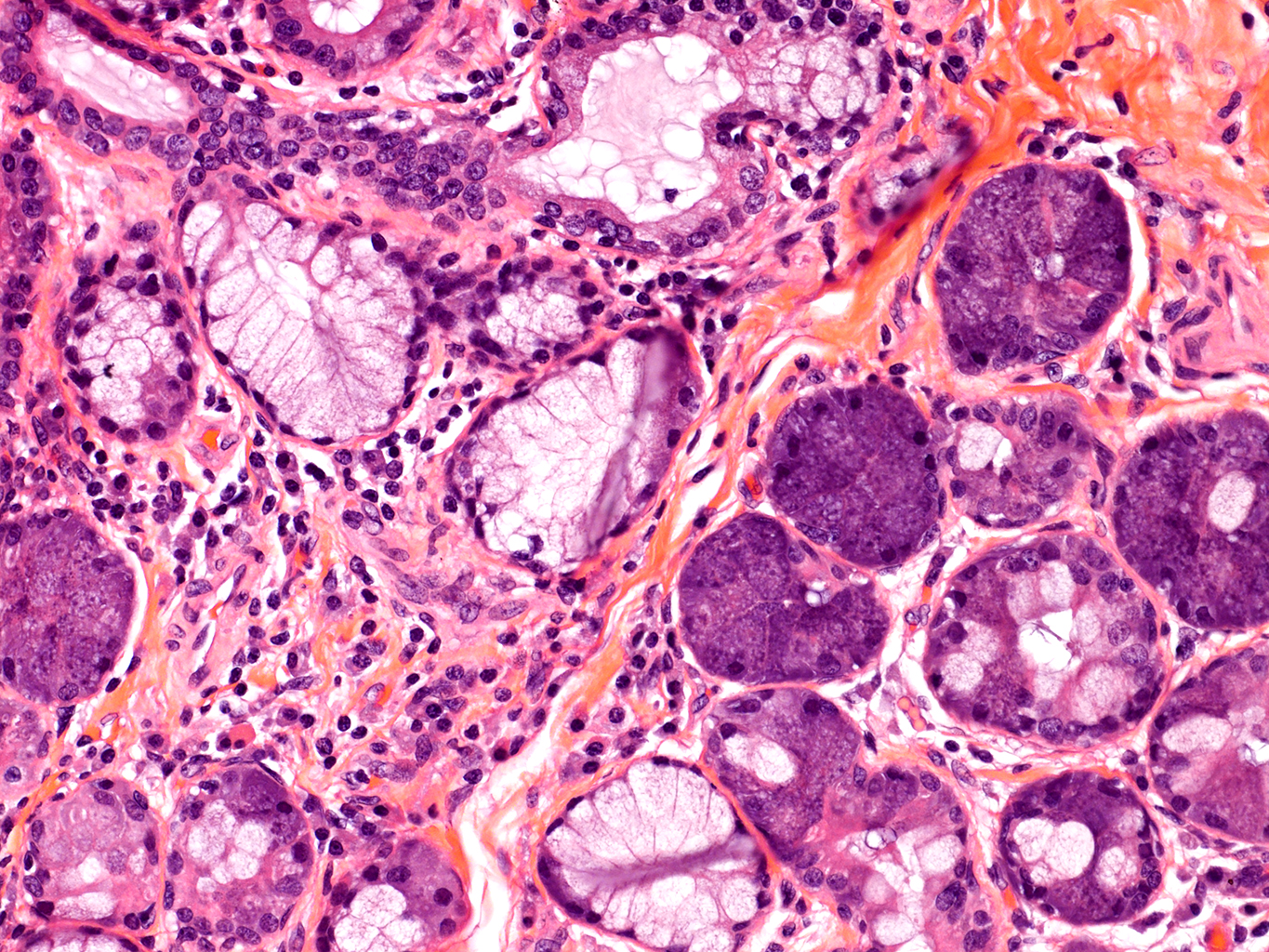
H&E Staining from PBR 3653 Patient with Cystic Fibrosis
20X
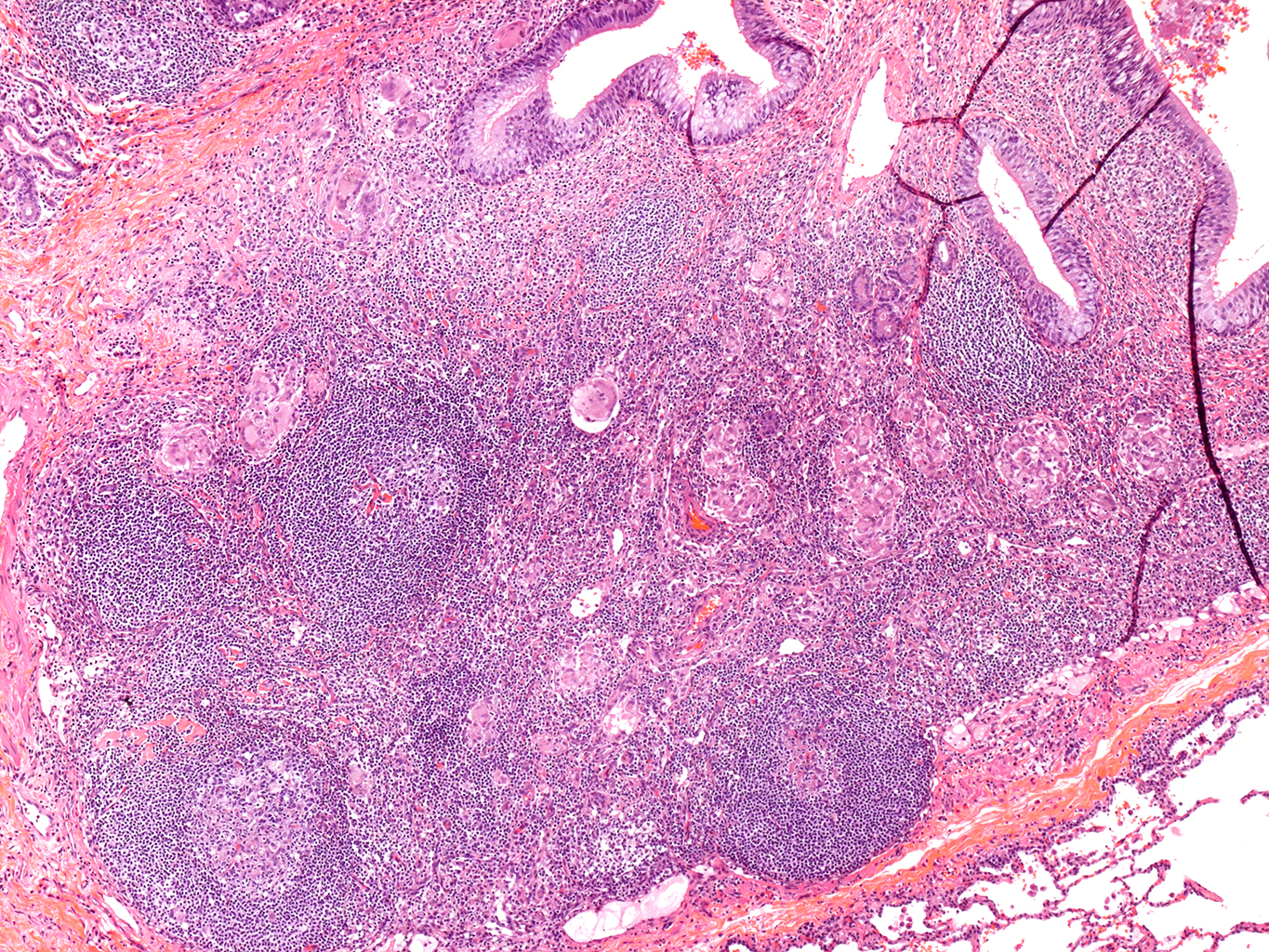
H&E Staining from PBR 3653 Patient with Cystic Fibrosis
4X
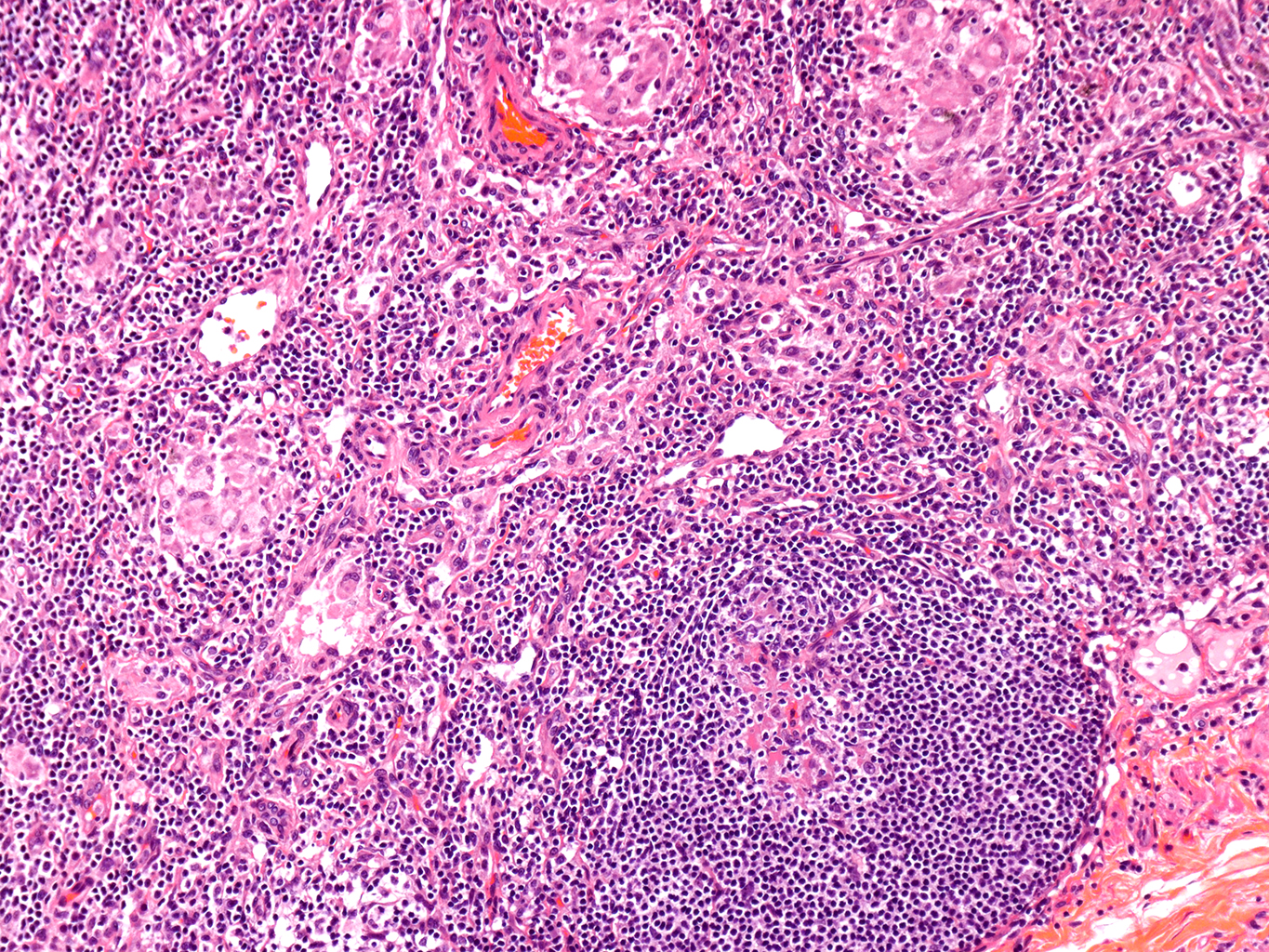
H&E Staining from PBR 3653 Patient with Cystic Fibrosis
10X
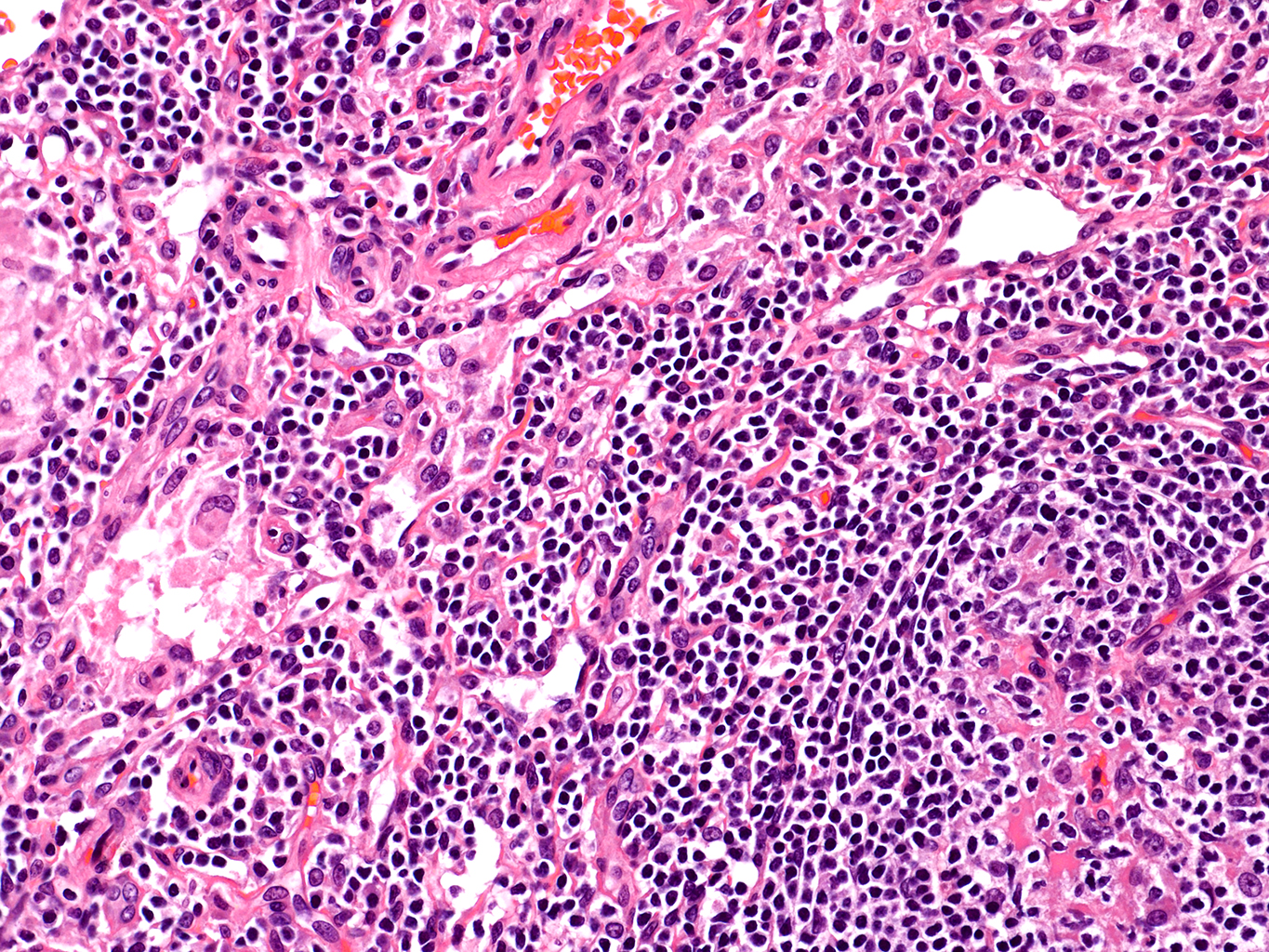
H&E Staining from PBR 3653 Patient with Cystic Fibrosis
20X
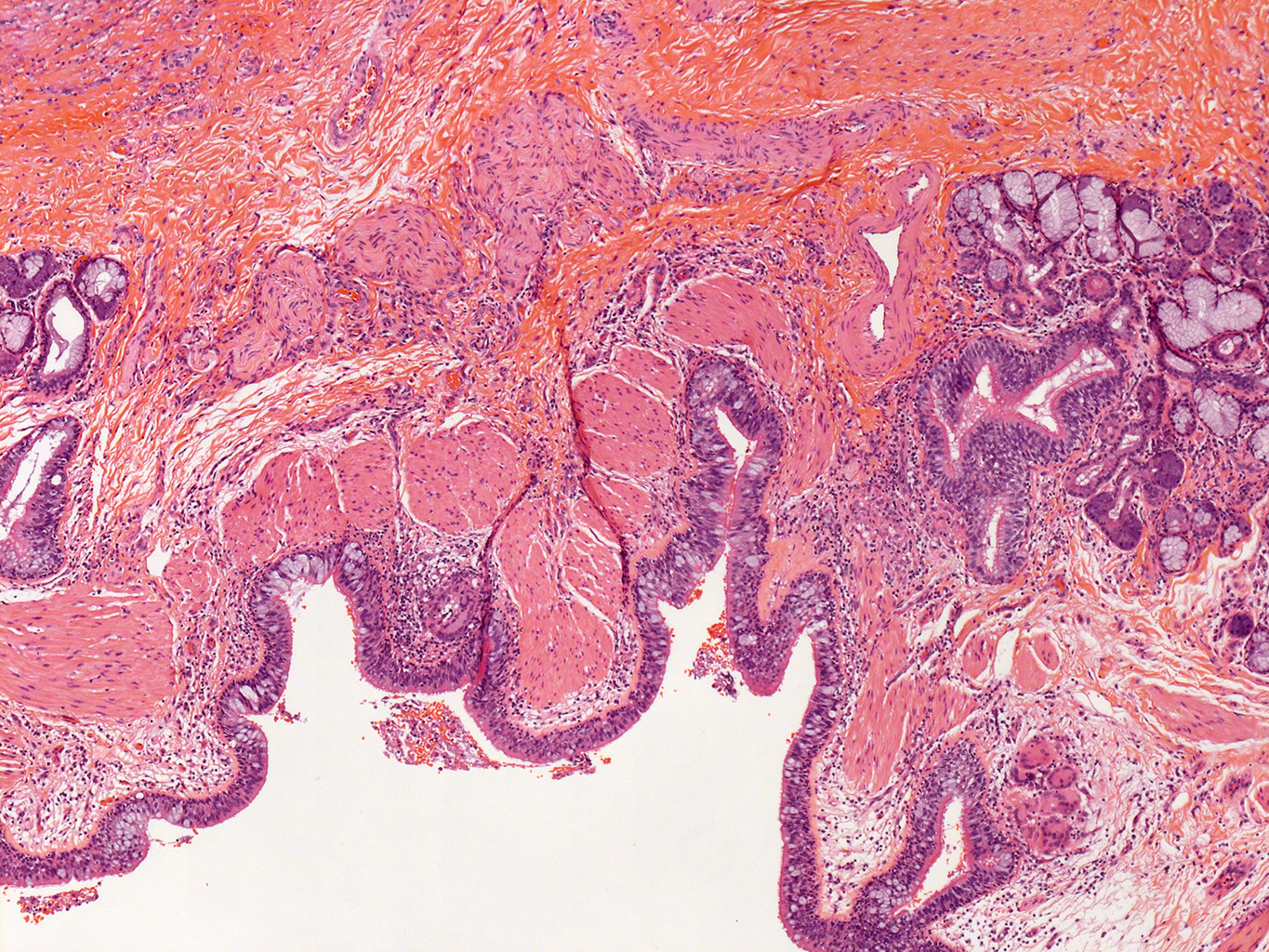
H&E Staining from PBR 3653 Patient with Cystic Fibrosis
4X
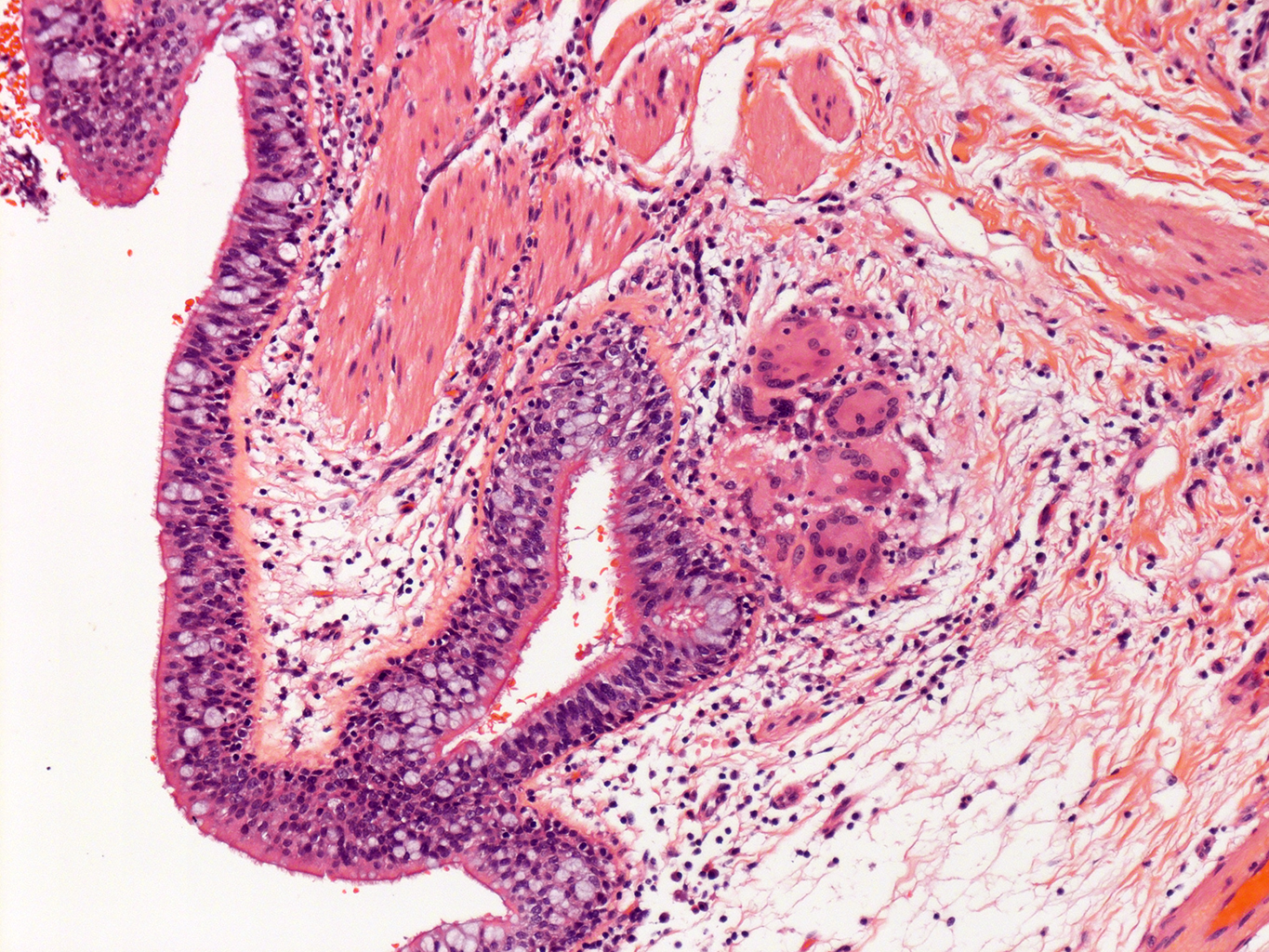
H&E Staining from PBR 3653 Patient with Cystic Fibrosis
10X
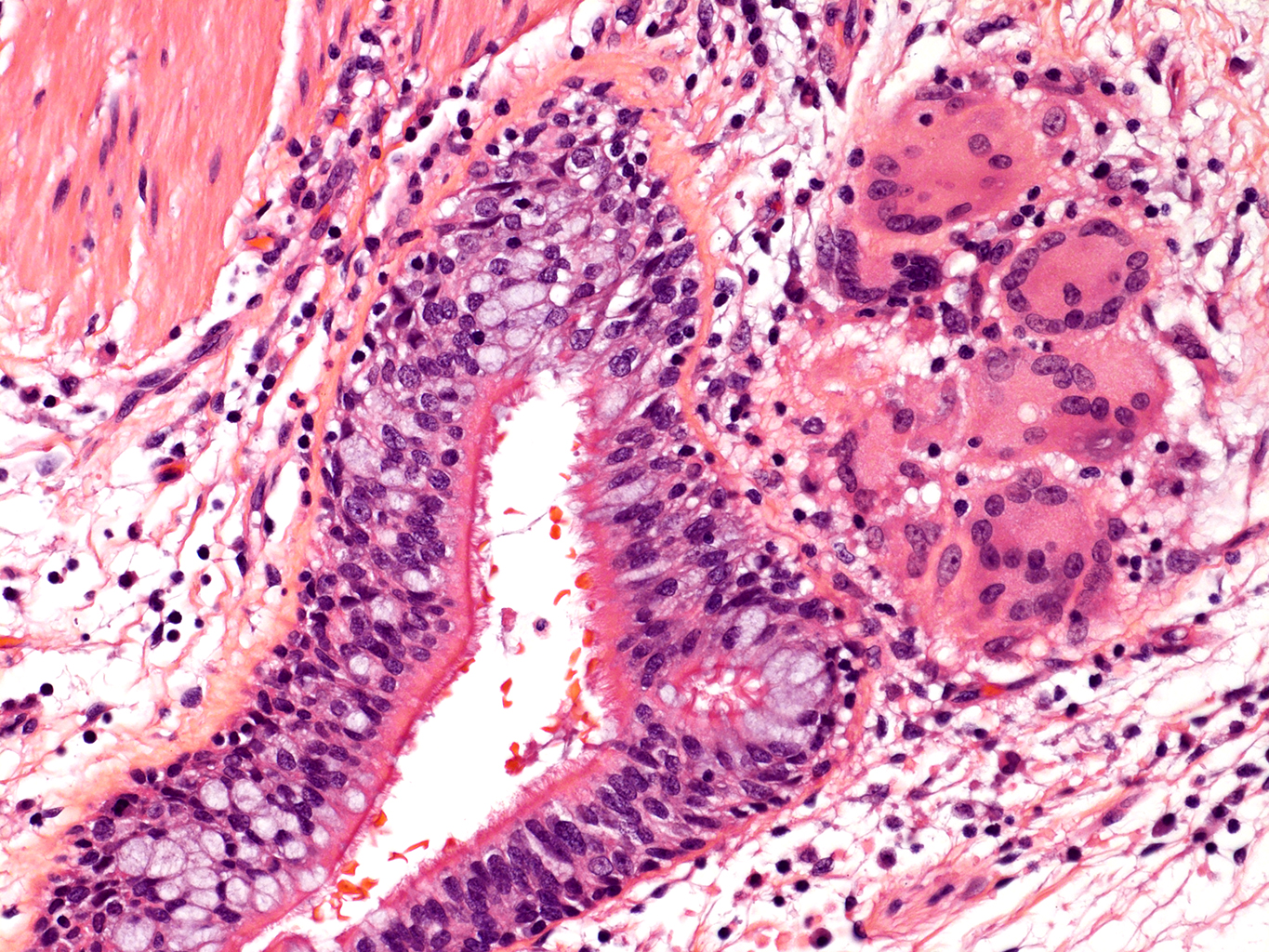
H&E Staining from PBR 3653 Patient with Cystic Fibrosis
20X
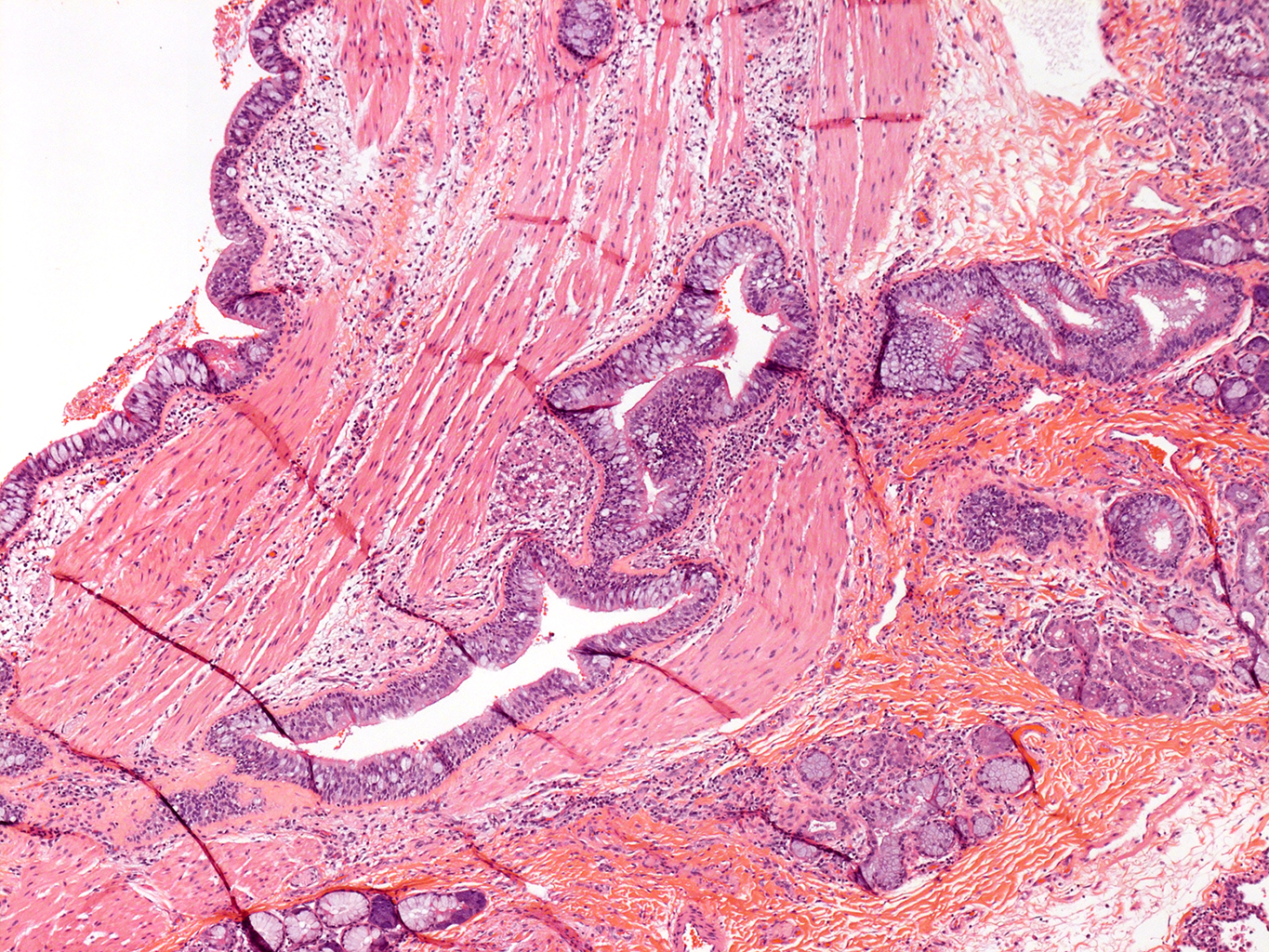
H&E Staining from PBR 3653 Patient with Cystic Fibrosis
4X
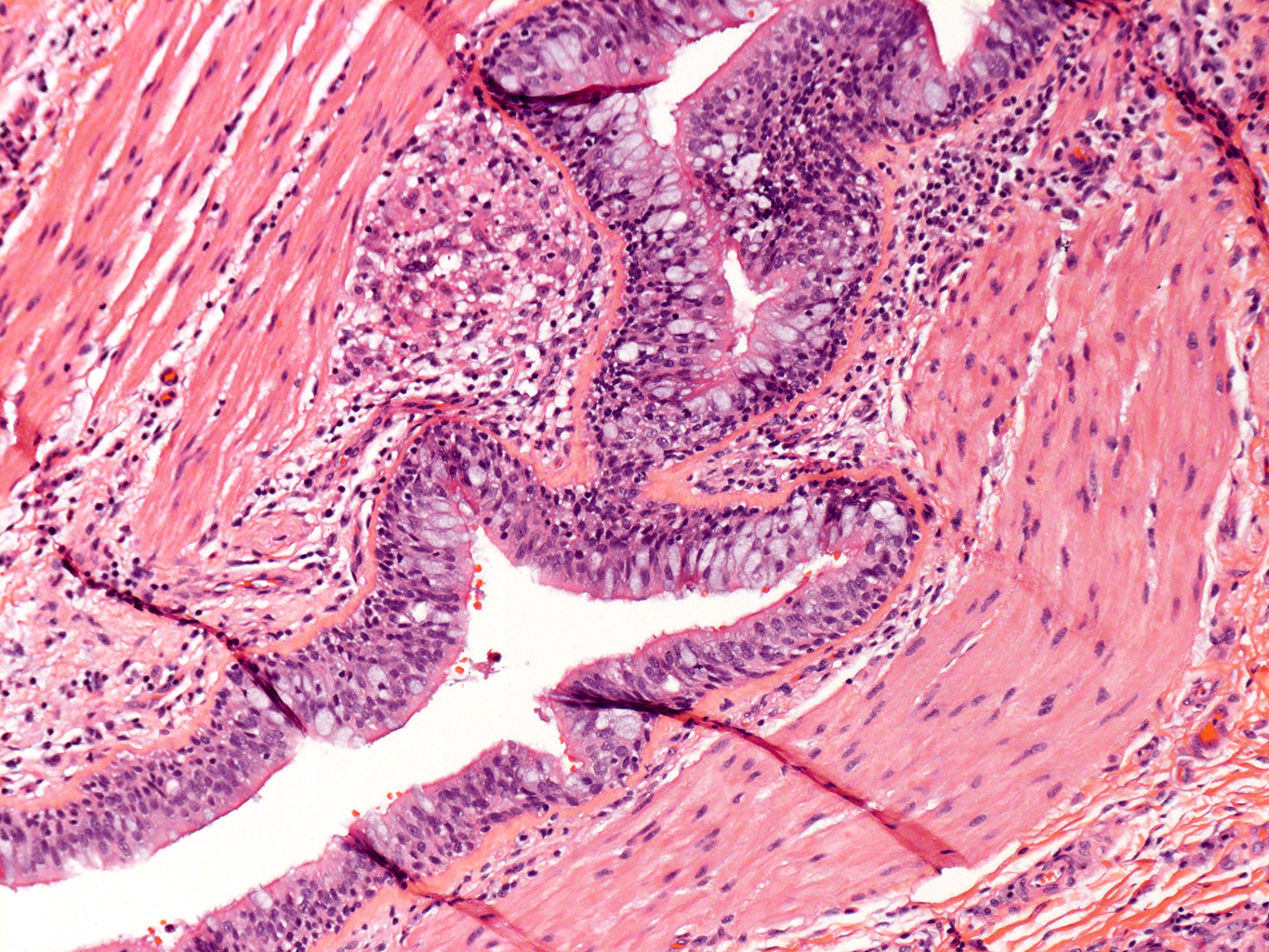
H&E Staining from PBR 3653 Patient with Cystic Fibrosis
10X
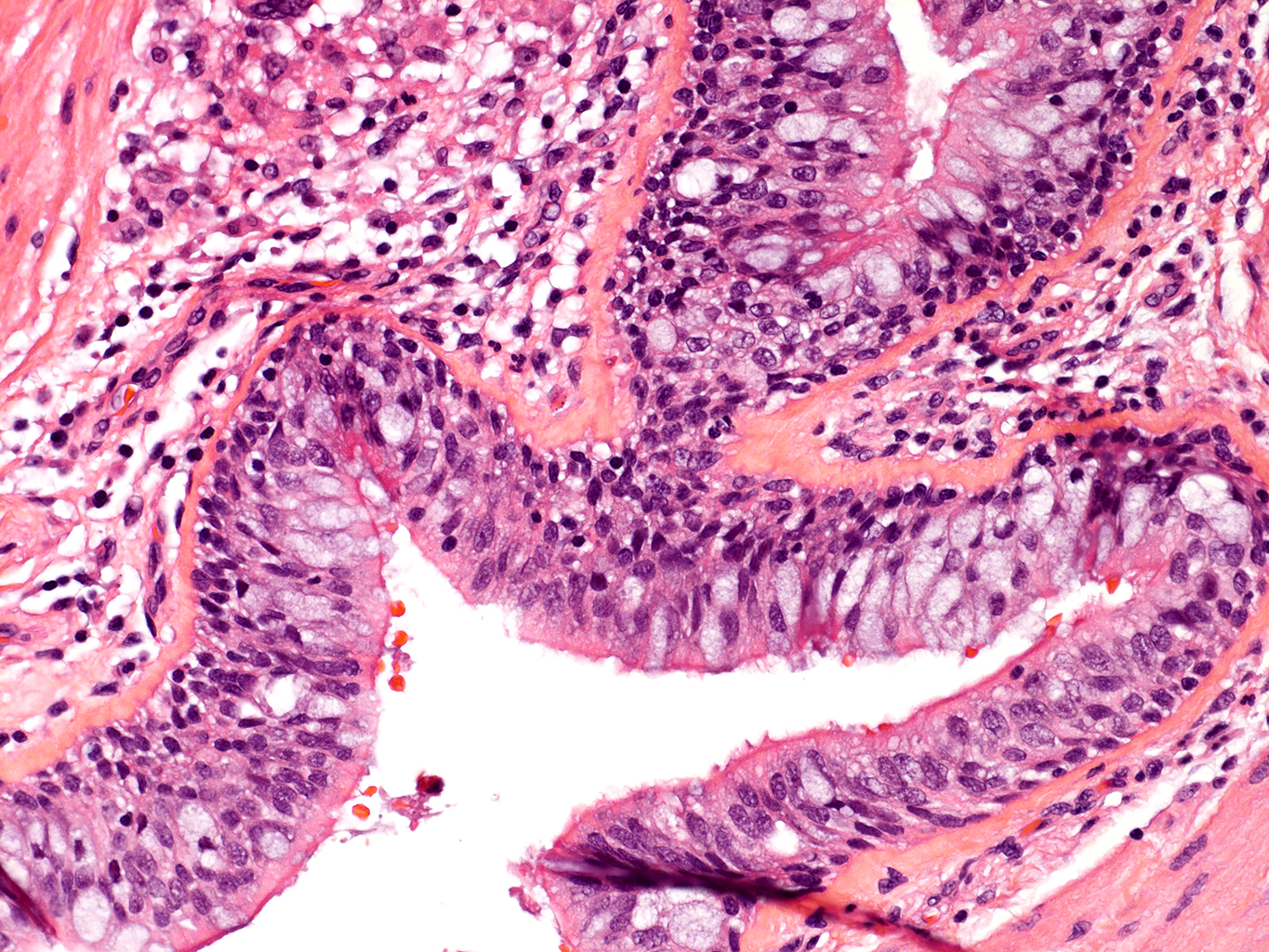
H&E Staining from PBR 3653 Patient with Cystic Fibrosis
20X
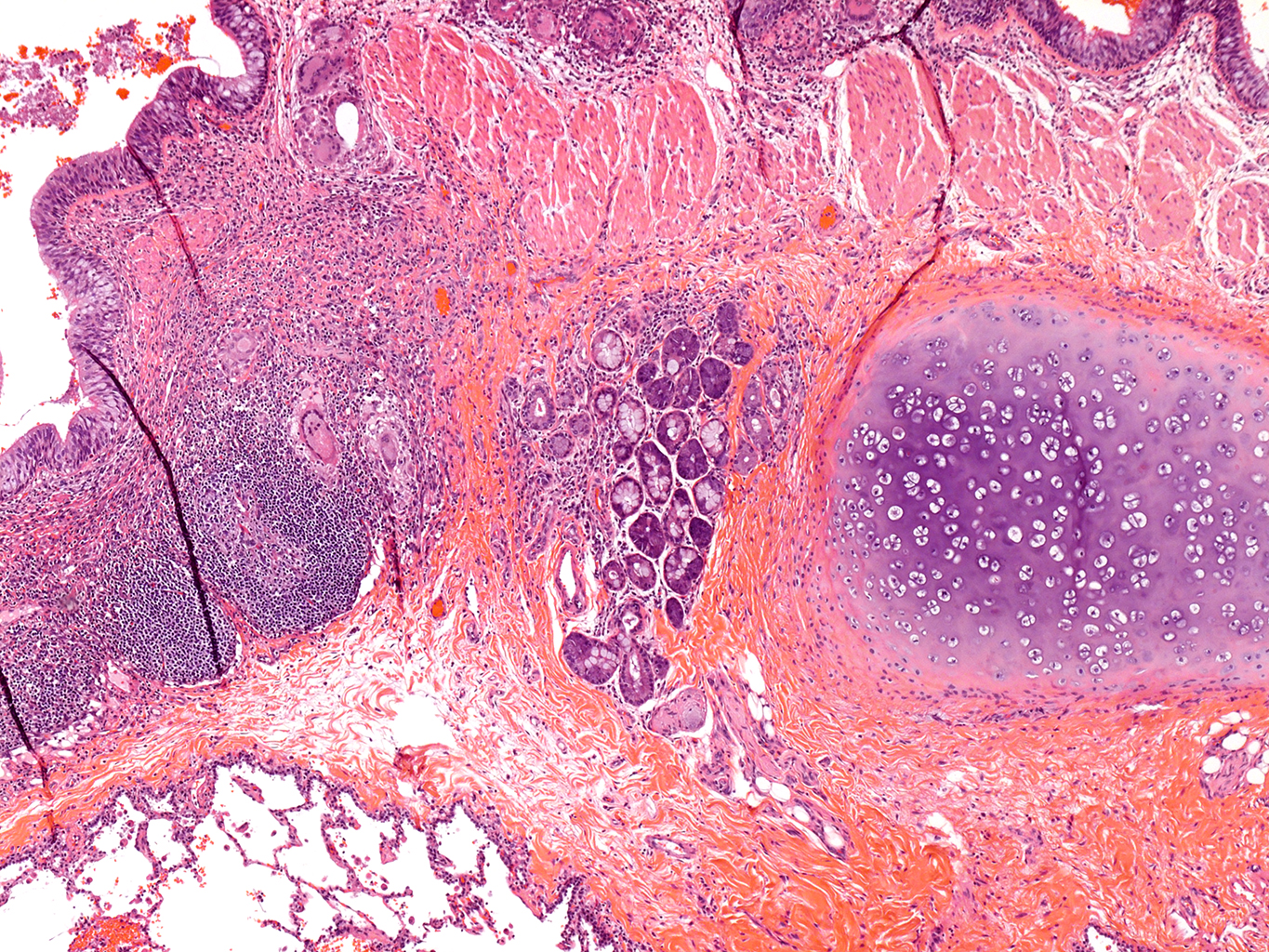
H&E Staining from PBR 3653 Patient with Cystic Fibrosis
4X
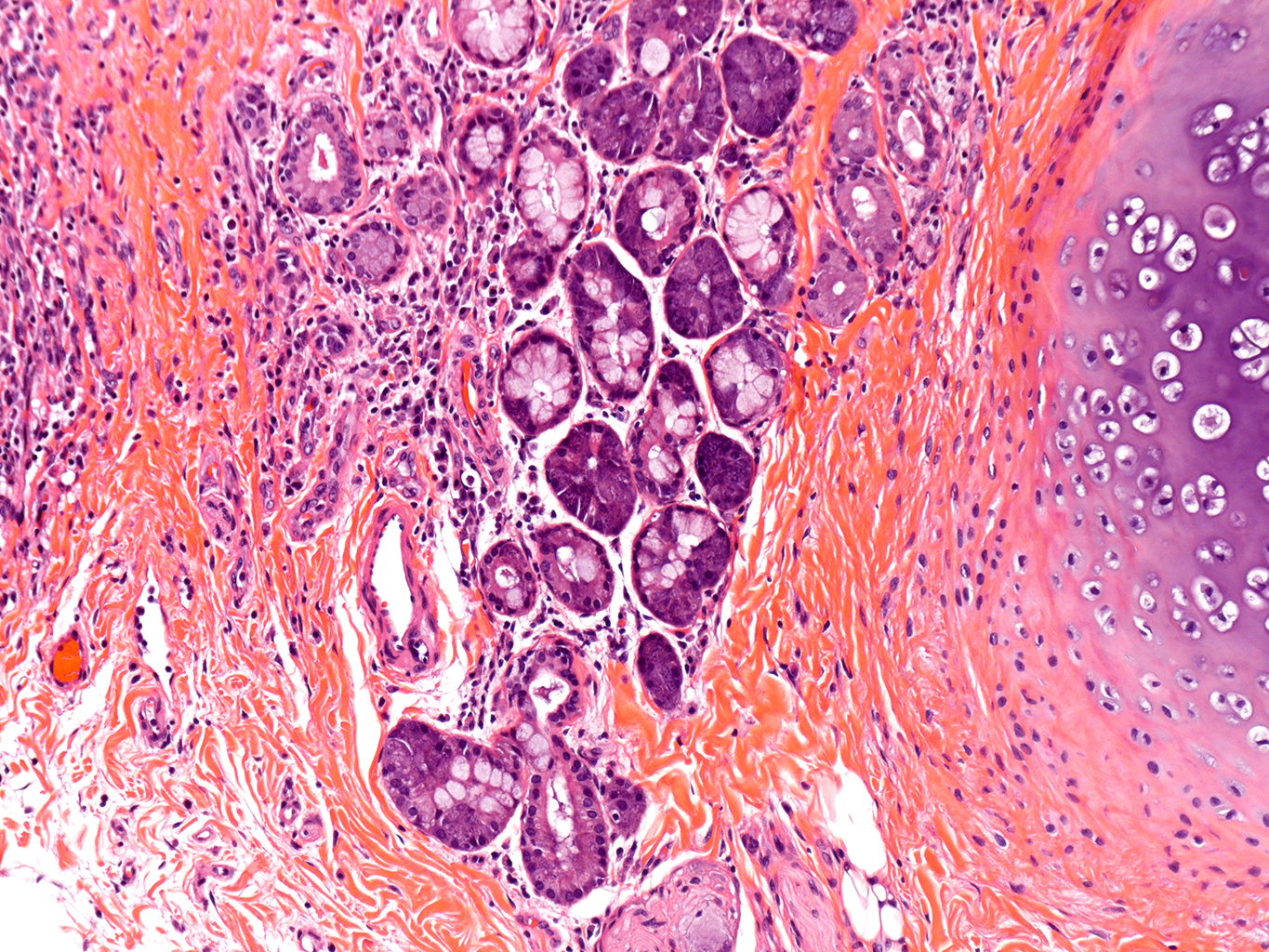
H&E Staining from PBR 3653 Patient with Cystic Fibrosis
10X
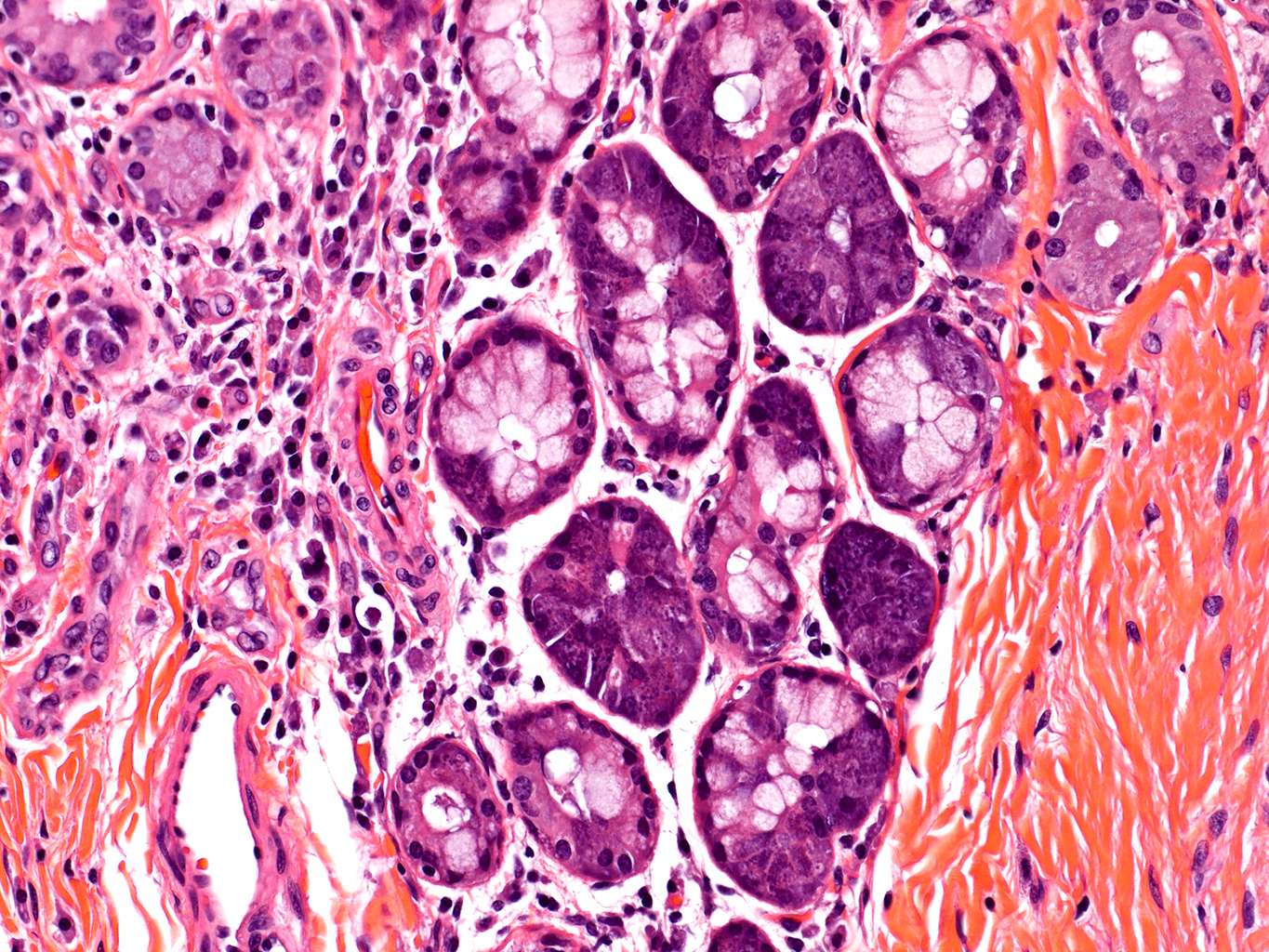
H&E Staining from PBR 3653 Patient with Cystic Fibrosis
20X
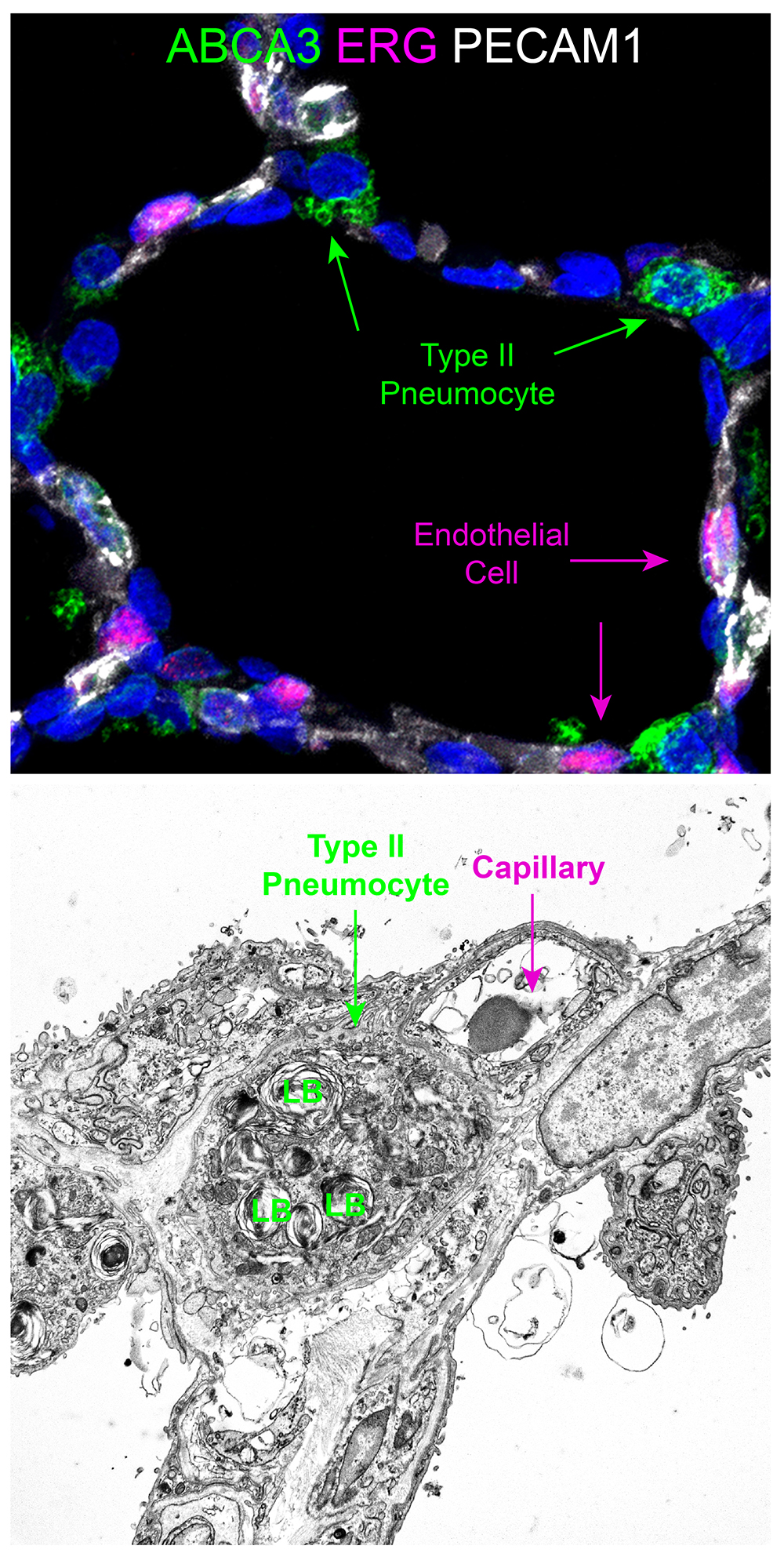
Donor Tissue Kindly Provided by Dr. Gloria Pryhuber from the University of Rochester Medical Center
- Paraffin sections were placed at 60oC overnight to melt paraffin.
- Paraffin sections were then placed in xylene (Xylenes Reagent Grade ACS Histology/Lab Grade Cat # 399000000 Pharma-AAPER) 3X for 10 minutes each.
- Rinsed slides in a series of 100% ETOH washes. 20 dips for the first wash, 3 minutes for the second wash, and 20 dips for the third wash.
- Rinsed slides in a series of 95% ETOH washes. 20 dips for the first wash, 3 minutes for the second wash, and 20 dips for the third wash.
- Rinsed slides in 70% ETOH for 3 min.
- Rinsed slides for 3 minutes in running dH2O.
- Slides were then stained in filtered Hematoxylin for 60 seconds. (Gold Standard Harris Hemotoxylin, Acidified, Mercury; Cat # 24245 Polysciences Inc,).
- Rinsed slides in tap water until water turned cleared then rinsed in dH2O
- Rinsed slides in Lithium Carbonate: 20 dips. (Lithium Carbonate ACS Grade; Cat # 255823-500G Sigma Aldrich). Lithium carbonate is prepared by over-saturating in dH2O.
- Rinsed slides in dH2O 3X 20 dips.
- Rinsed slides in 70% ETOH for 20 dips.
- Rinsed slides in 80% ETOH for 20 dips.
- Stained slides in Eosin for 15 seconds. (Eosin Y 0.5% Alcohol Solution, Cat # 09859 Polysciences Inc,).
- Rinsed slides in a series of 95% ETOH washes. 10 dips for the first and second washes, 15 dips in the third and fourth washes.
- Rinsed slides 3X in 100% ETOH. 10 dips for the first and second washes and 15 dips in the third and fourth washes.
- Rinsed slides in 2X in Xylene, followed by 6 more washes in xylene.
- Coverslip with Permount. (Permount Toulene Solution UN 1294, Cat # SP-15-500 Fisher Scientific).
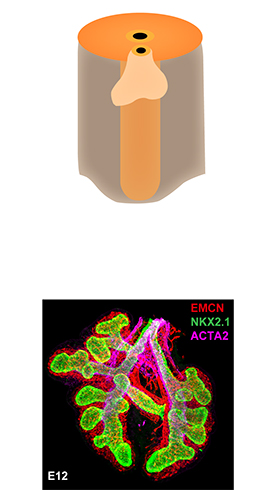
Tissue Used:
PBR 3653 Patient with Cystic Fibrosis