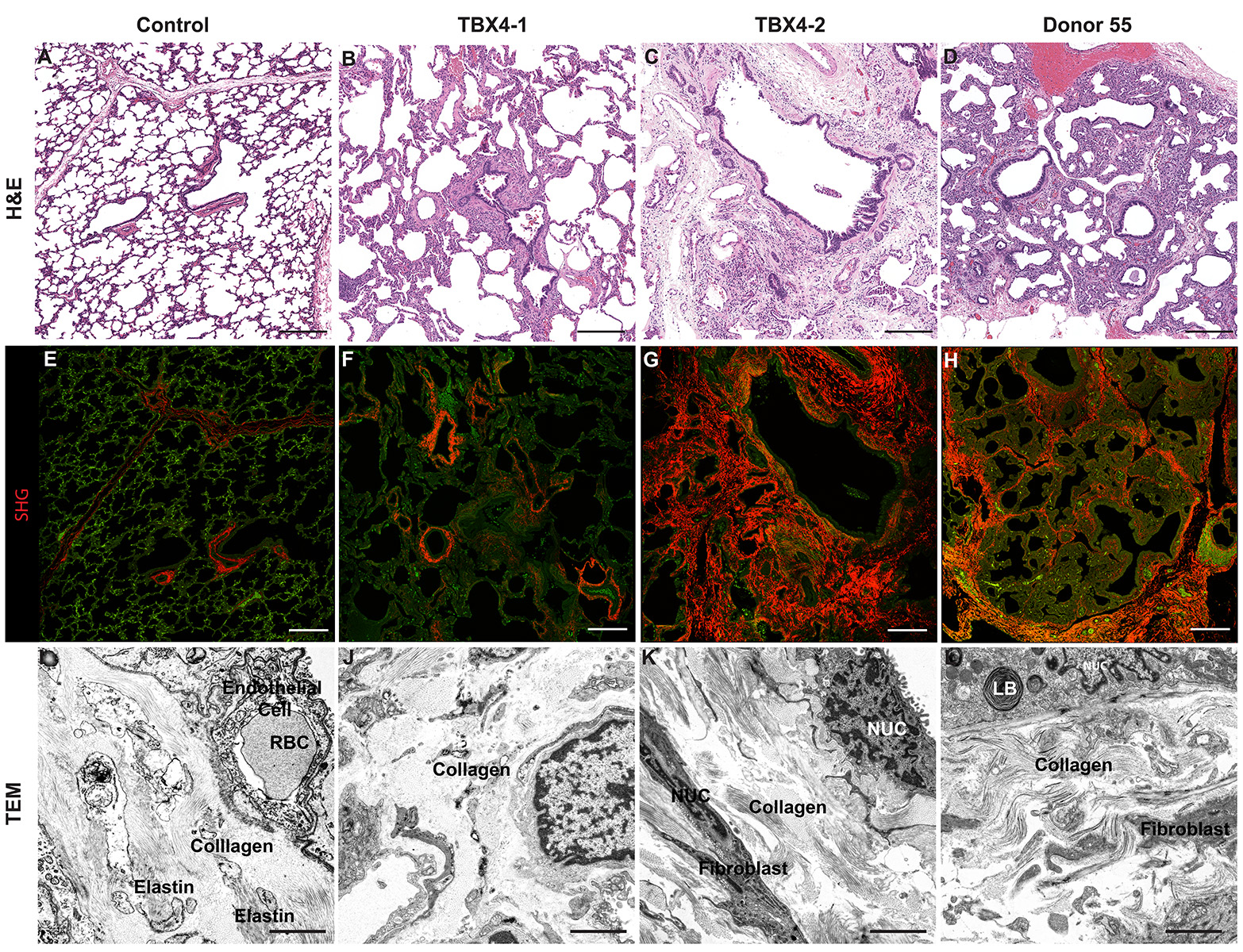
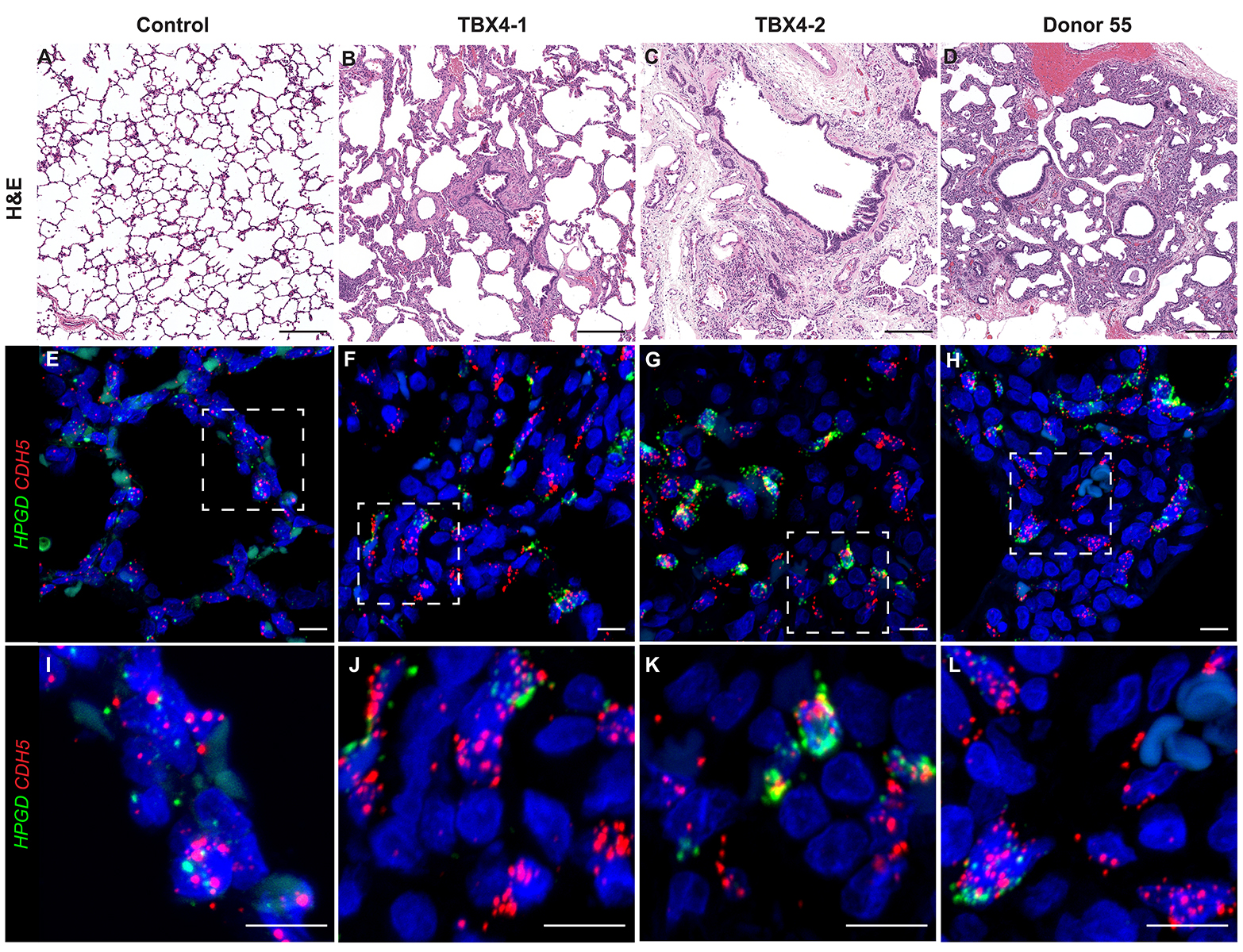
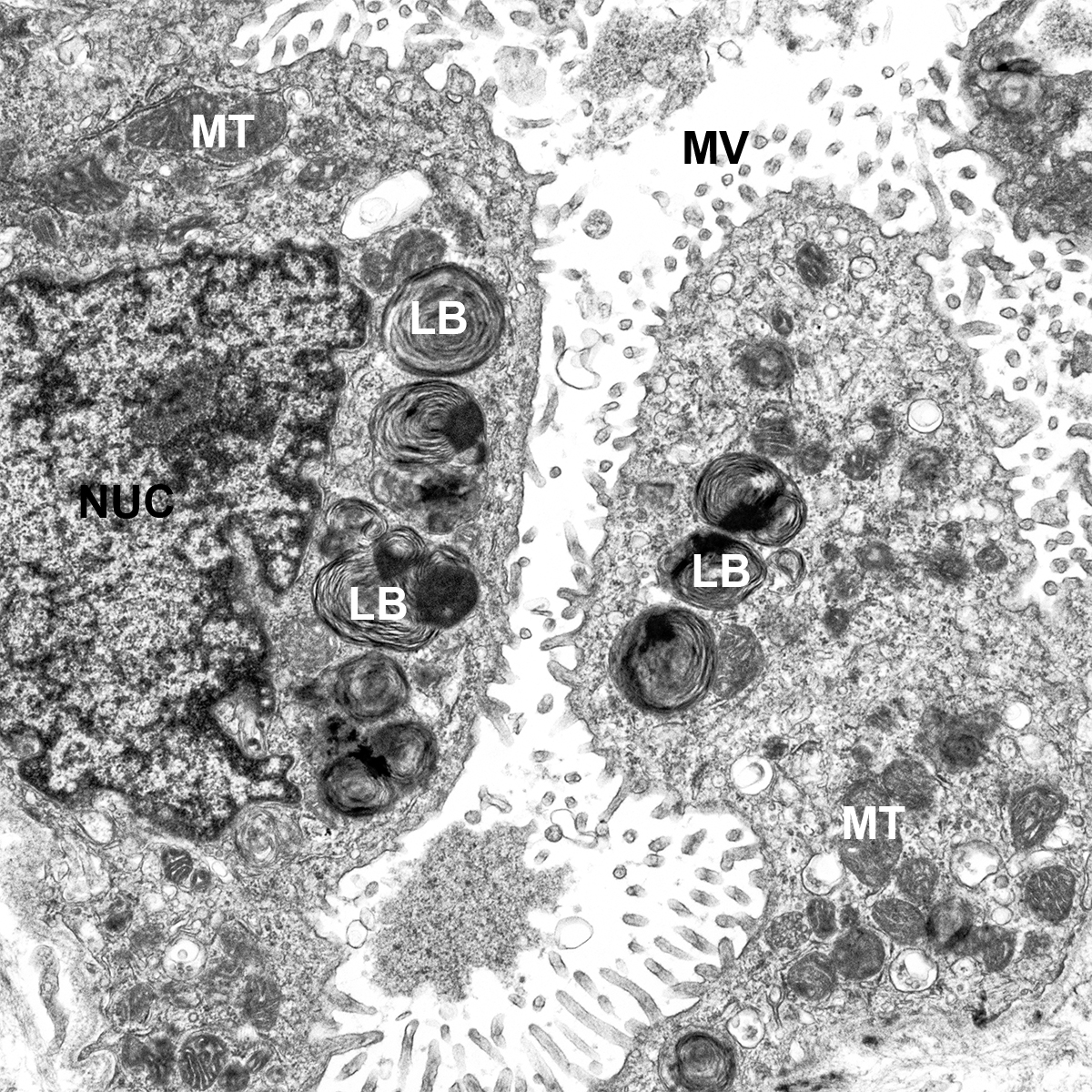
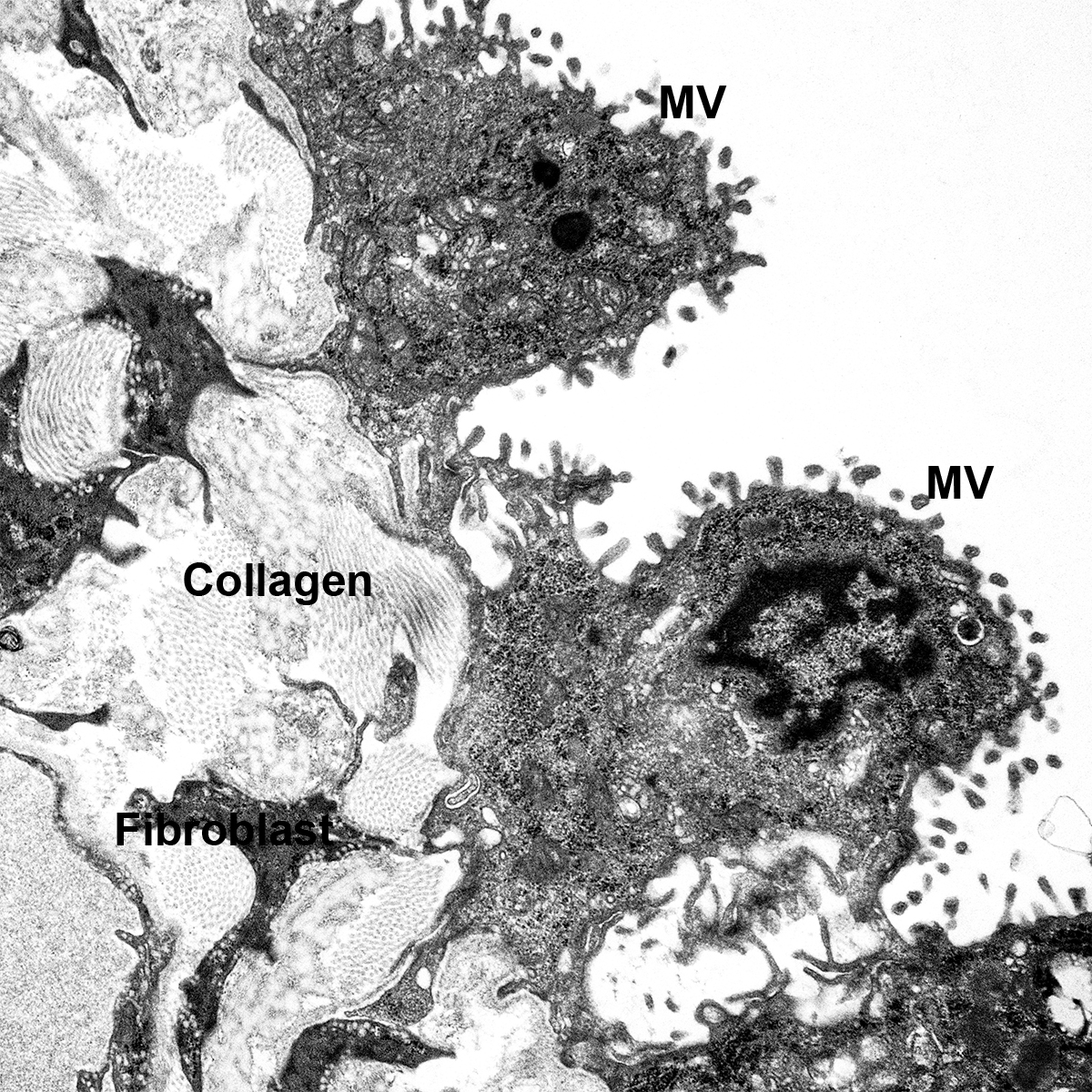
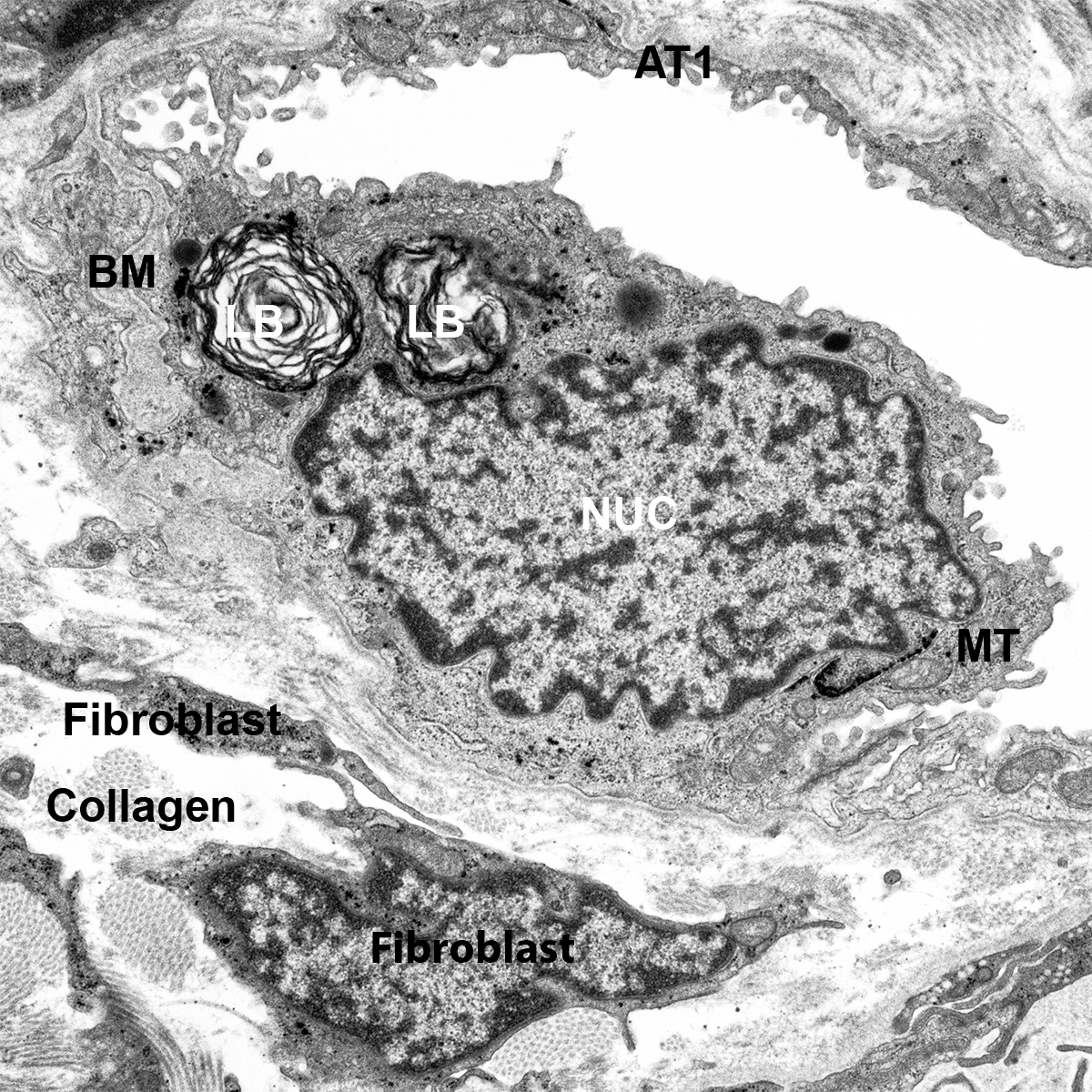
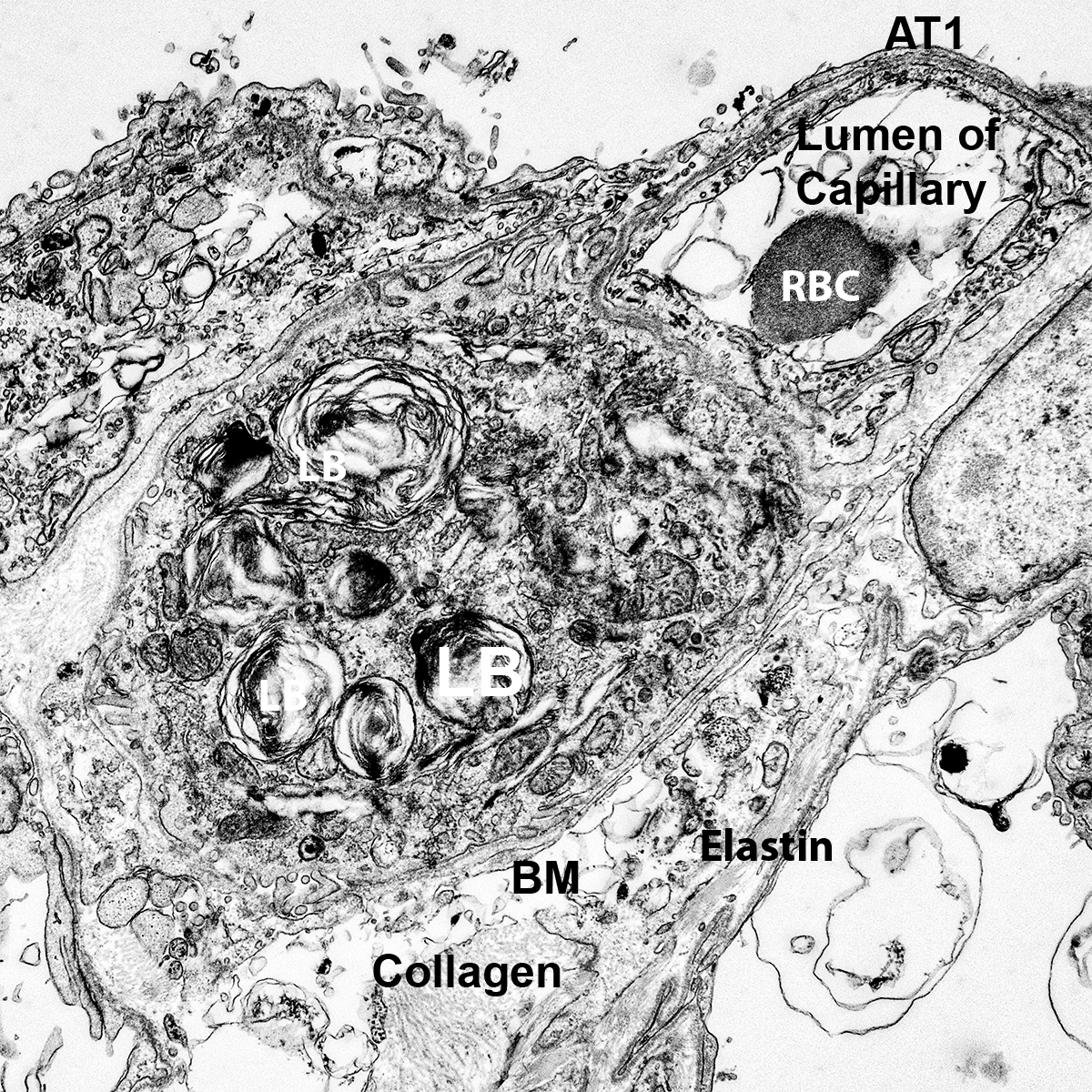
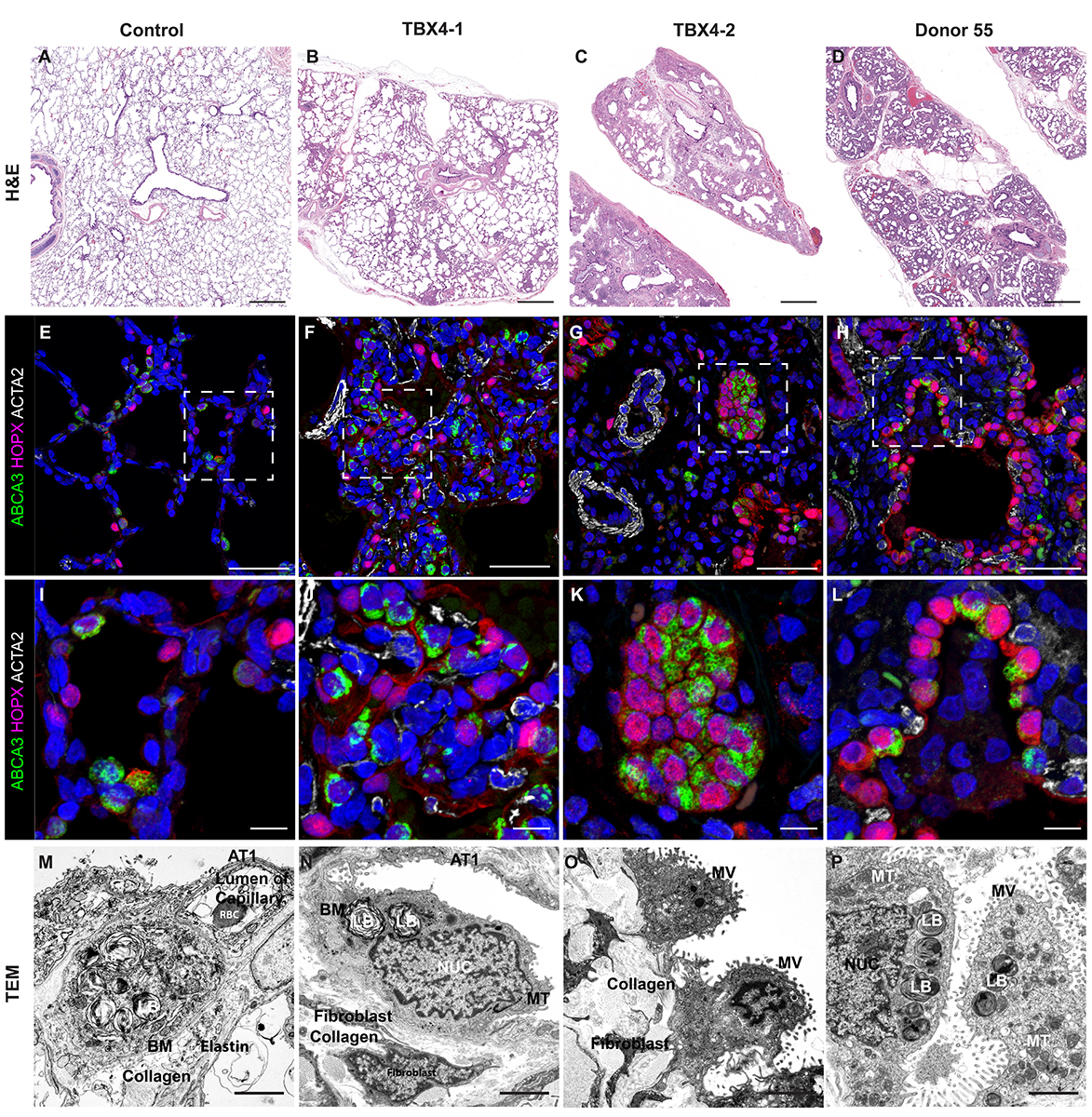
CCHMC Mouse SOP Timed Matings and Tissue Harvest, Fixation, and Processing
CCHMC Mouse SOP: Timed Matings & Tissue Harvest/Fixation/Processing
A. MICE
1. C57BL/6J are obtained or derived from the Jackson Laboratory colony.
2. Breeding females should be between 8-15 weeks of age and no older than 6 months.
3. Male mice can be used up to 7-8 months of age, or until no longer producing pregnancies.
4. Expect seasonal changes in breeding performance. Some strains produce more litters in the spring and summer than in winter and fall.
B. BREEDING
1. Females should have one litter before setting up breedings for experimental material. NOTE: First litters may not be representative of the average number of pups per litter for the strain.
2. Place 1 to 3 females into each male’s cage in the late afternoon, prior to the dark-cycle. Record date and time.
3. Check for vaginal plugs early the next morning (plug will dissolve over time).
4. By convention, the date a plug is observed is designated as gestational day 0 or 0.5. We use 0.5 (or E0.5). Record date and time.
5. Remove male from cage.
6. Separate unplugged female mice from the male. Do not use these female mice for breeding until it can be demonstrated that there is no pregnancy. This can be done by monitoring daily and/or by palpitation.
C. HARVESTING
1. Harvest embryos in the morning (before noon) on the desired gestational day.
2. Obtain fetuses by hysterectomy. Dam should be anesthetized and then exsanguinated to prevent congestion of blood in the tissues.
3. Remove pups from uterus and maintain in a large weigh boat on ice. As the pups are extracted from the uterus, place each pup in a smaller weigh boat on ice, and number or label for future identification.
4. Strip way extra-fetal membranes.
5. Weigh and measure crown-to-rump (C-R) length for each pup prior to fixation and record. NOTE: Variations in lung development within a litter are directly correlated with fetal body weight and crown-to-rump length measurements.
6. Compare body weights and C-R lengths with fetal growth tables to determine developmental age. See attached or below.
7. Remove a piece of tail or tissue for isolating genomic DNA.
D. FIXATION AND PROCESSING
1. For E12.5 to E14.5, leave embryos intact and immerse in 20 ml of 4% PFA in 1XPBS (formulated with DEPC-treated water) in a 50 ml sterile tissue culture tube. Fix at room temperature for one hour, and then at 4oC overnight. Number/label tubes accordingly. NOTE: Volume of fixative should be at least 10-20 times the volume of the tissue. Container should be at least 2-3 times the diameter of the tissue. Fixative should cover fetus
2. Larger pups between E16 and postnatal day 5 should be kept on ice as above, weighed and measured, and then exsanguinated.
3. Open the abdomen and clip the descending aorta and/or vena cava to let the animal bleed out.
4. Divide the pup into two segments with a sharp razor blade at the level of the diaphragm, cutting just below the inferior extent of the thorax or below the liver.
· Remove liver and diaphragm, and trim rib cage. NOTE: You must strip off the diaphragm prior to immersion in the fixative in order to allow the fixative to enter the chest cavity directly.
· If leaving the chest intact, the head may be removed by bisecting it between the upper and lower jaws. The lower jaw will still be attached to the neck. This prevents the larynx and trachea from retracting.
· Alternately, remove lung from the chest.
· Place in 20 ml of 4% PFA/1X PBS, incubate at room temperature for 1 hour, and then incubate at 4oC overnight. Number/label containers accordingly. NOTE: If the fixative becomes bloody from the initial dissection, then rinse and replace with fresh fixative.
3. Wash 3 times in 1X PBS (formulated with DEPC-treated water) for 10 minutes each at room temperature.
4. Incubate in 30% Sucrose in 1X PBS (formulated with DEPC-treated water) at 4oC.
5. The day before freezing place in 2 parts 30% Sucrose/1XPBS and 1 part OCT, and incubate at 4oC.
6. Freeze in molds filled with OCT on dry ice or in isopentane that has been pre-chilled in a container surrounded by dry ice.
7. Alternately, embryos can be prepared for paraffin embedding. Following step 3, the tissue is dehydrated through 30% and 50% ETOH for 10 minutes each, and then 70% ETOH, 3 times, 10 minutes each. All solutions are prepared with DEPC-treated water. Tissue can then be stored in 70% ETOH at 4oC until processed for paraffin embedding.
8. Embed each fetus so that it is resting on its back with the heart facing up. When embedding E18.5, lift up the fetus so the fetus is resting more on the shoulder blade.
E. RNA EXTRACTION
1. Alternatively, tissue can be harvested for RNA extraction. Extract the lungs from the chest and place in 1X PBS.
2. Carefully, dissect each lobe away from the heart, trachea, bronchi, esophagus.
3. Place in collection tube and immediately place on dry ice. Store at -80oC until RNA can be isolated.
F. SOLUTIONS FOR FIXATION / PROCESSING
1. PBS (phosphate-buffered saline)
10X stock solution, 1 liter, pH 7.2 to 7.4* 20X stock solution
80 g NaCL 160 g NaCl
2 g KCl 4 g KCL
11.5 g Na2HPO4-7H2O 23 g Na2HPO4-7H2O
2 g KH2PO4 4 g KH2PO4
2. 4% paraformaldehyde in PBS
Must be made up fresh, i.e. on the same day as fixation.
(a) Recipe #1 – for small volumes.
10 ml of 16% paraformaldehyde stock solution, EM grade*
4 ml of 10X PBS (prepared as above)
26 ml of D2H2O
40 ml total (pH 7.2-7.4)
(b) Recipe #2 – for larger volumes.
Add 8 g to 100 ml of water (ddH2O). Heat to 60°C in a fume hood. Add a few drops of 1 N NaOH to help dissolve. When the solid has completely dissolved, let the solution cool to room temperature, and add 100 ml of 2X PBS and adjust pH to 7.4. This solution should be prepared fresh. You may have to filter solution if a precipitate persists. We have also frozen batches of this solution at -20°C for subsequent use.
These are basic recipes. You will have to make more if fixing a large number of animals. Figure 20 ml per fetus (or 10X the volume of the specimen). You may fix the tissue in sterile 50 ml tissue culture tubes.
*obtain from Electron Microscopy Sciences, #15710,10 X 10 ml per box, 1-800-523-5874.
G. Gender Identification PCR
http://www.karger.com/Article/FullText/348677
McFarlane L, Truong V, Palmer J, S, Wilhelm D, Novel PCR Assay for Determining the Genetic Sex of Mice. Sex Dev 2013;7:207-211
SXF 5ʹ-GATGATTTGAGTGGAAATGTGAGGTA-3ʹ
SXR, 5ʹ-CTTATGTTTATAGGCATGCACCATGTA-3ʹ
PCR Conditions
94oC 2 minutes
94oC
57oC 30 Seconds, 35 cycles
72oC
72oC 5 minutes
4oC Hold