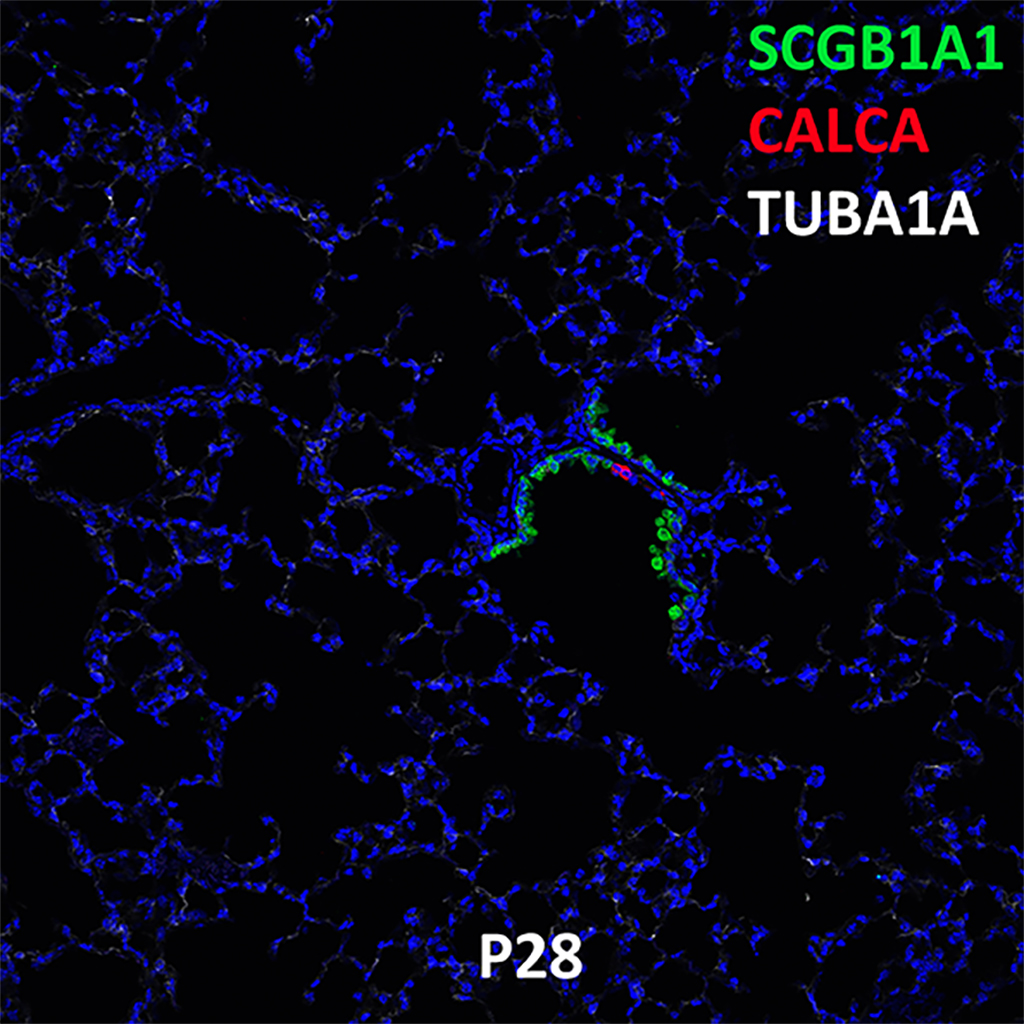
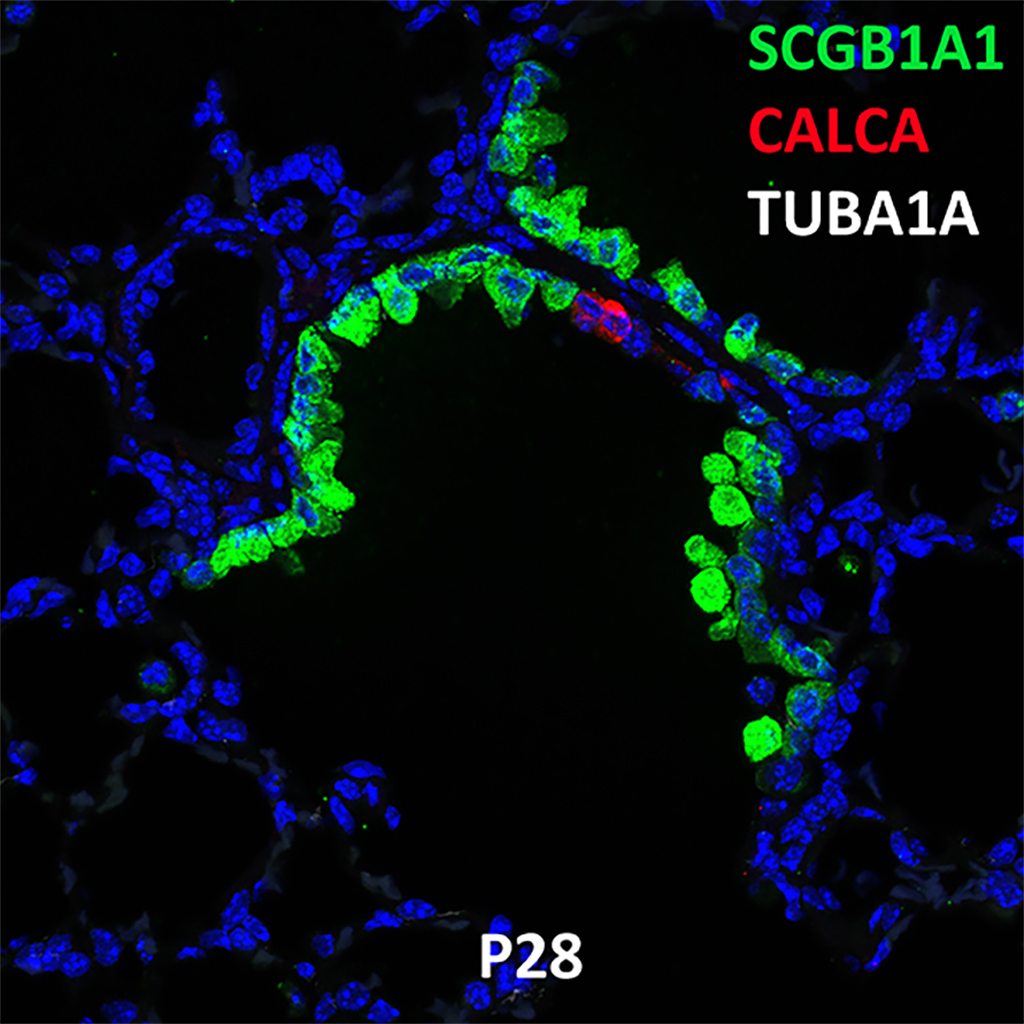
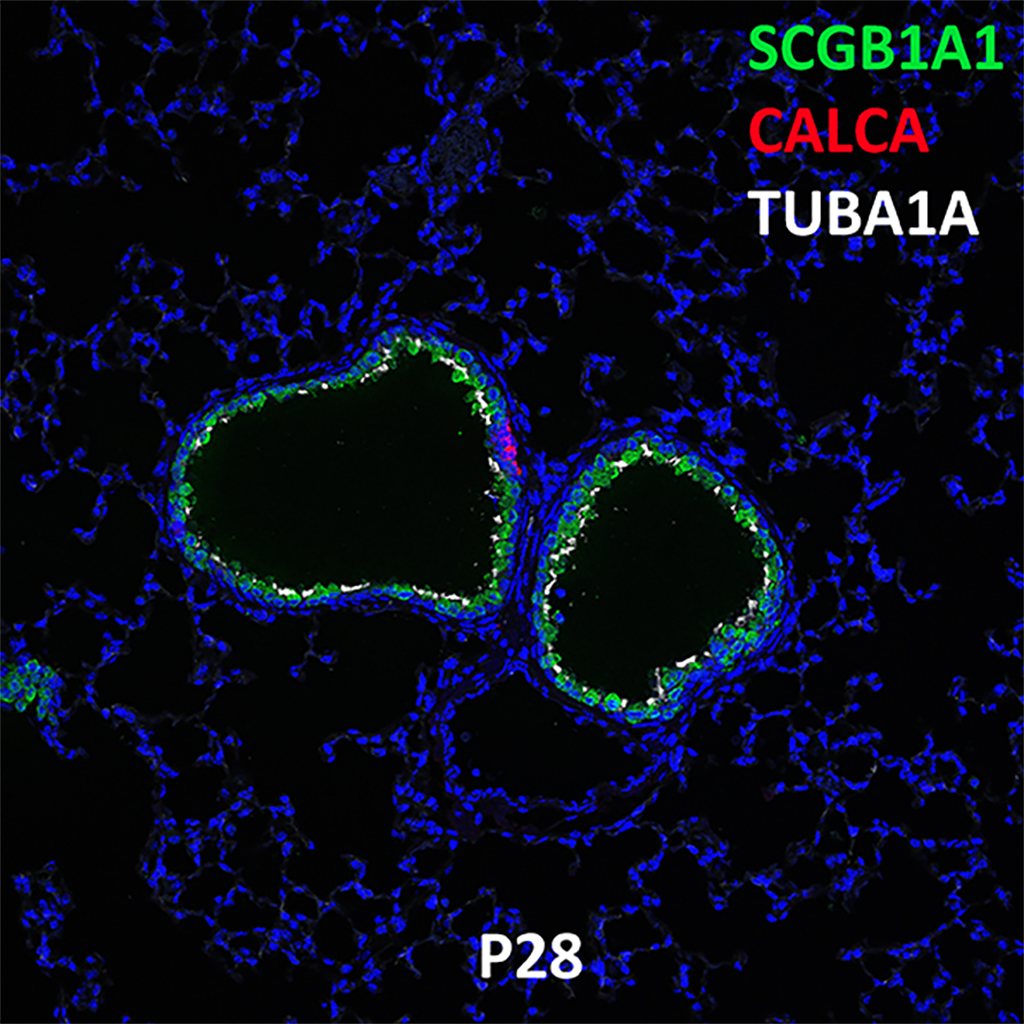
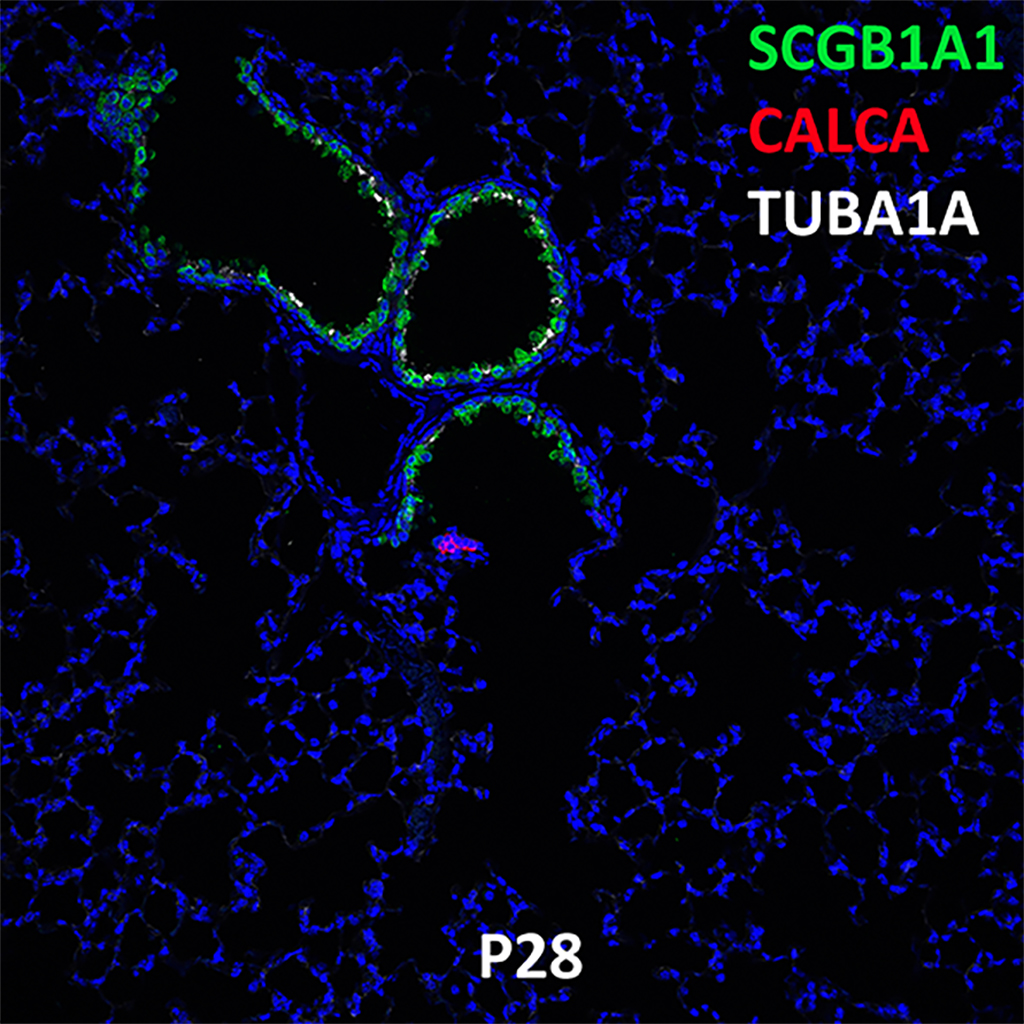
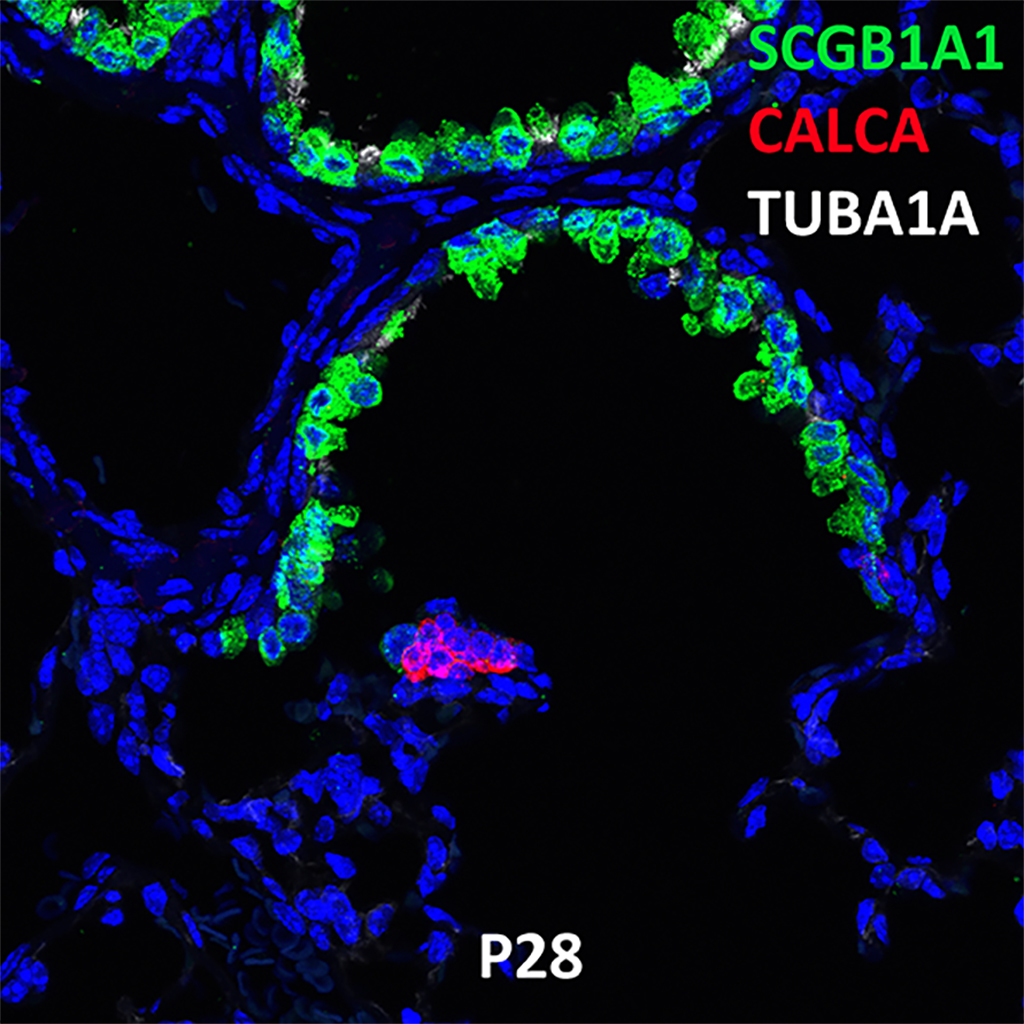
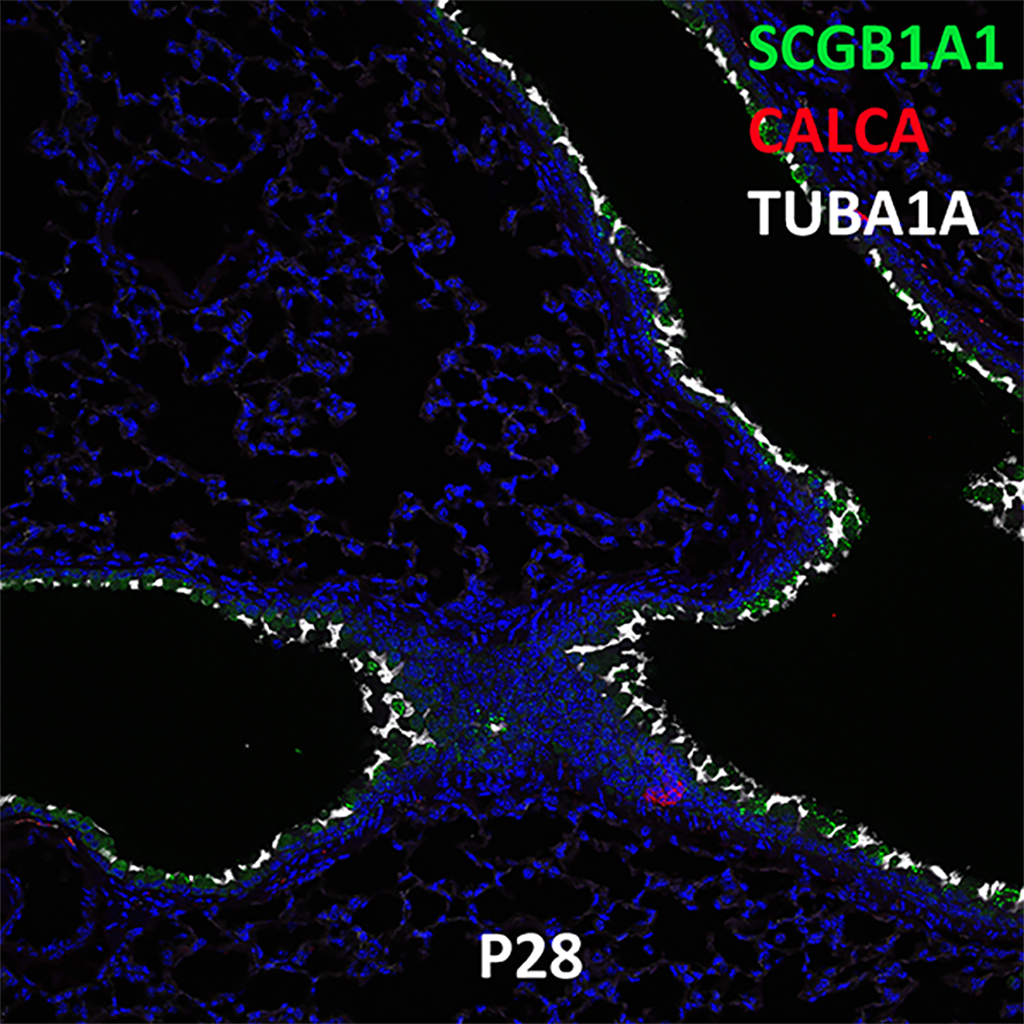
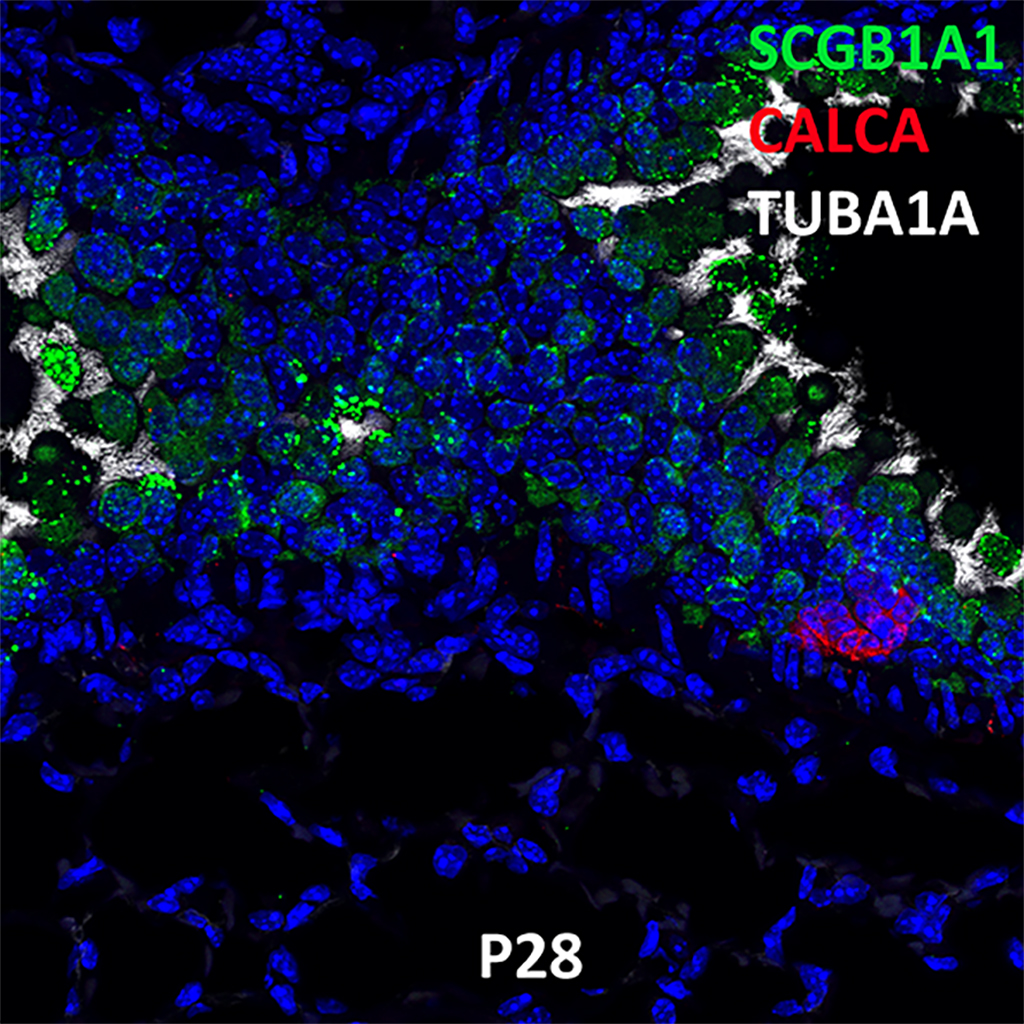
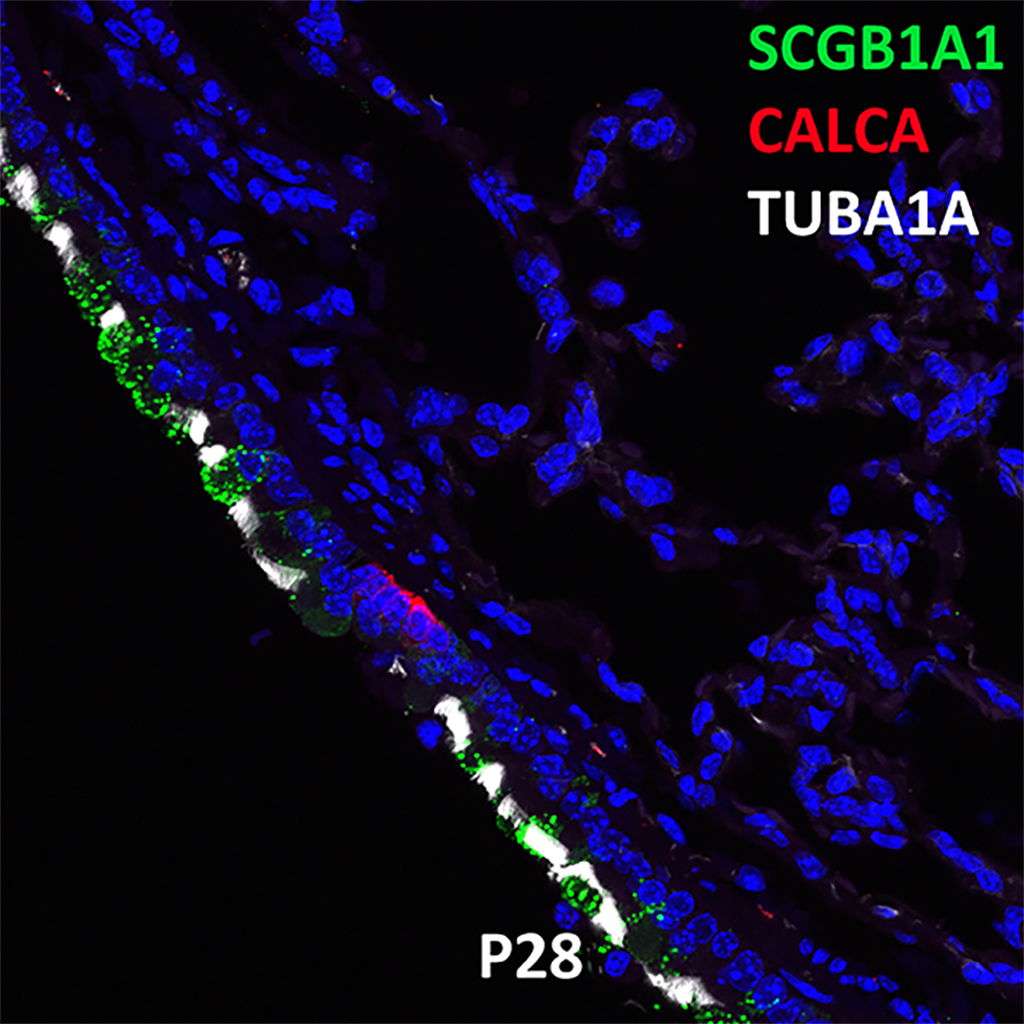
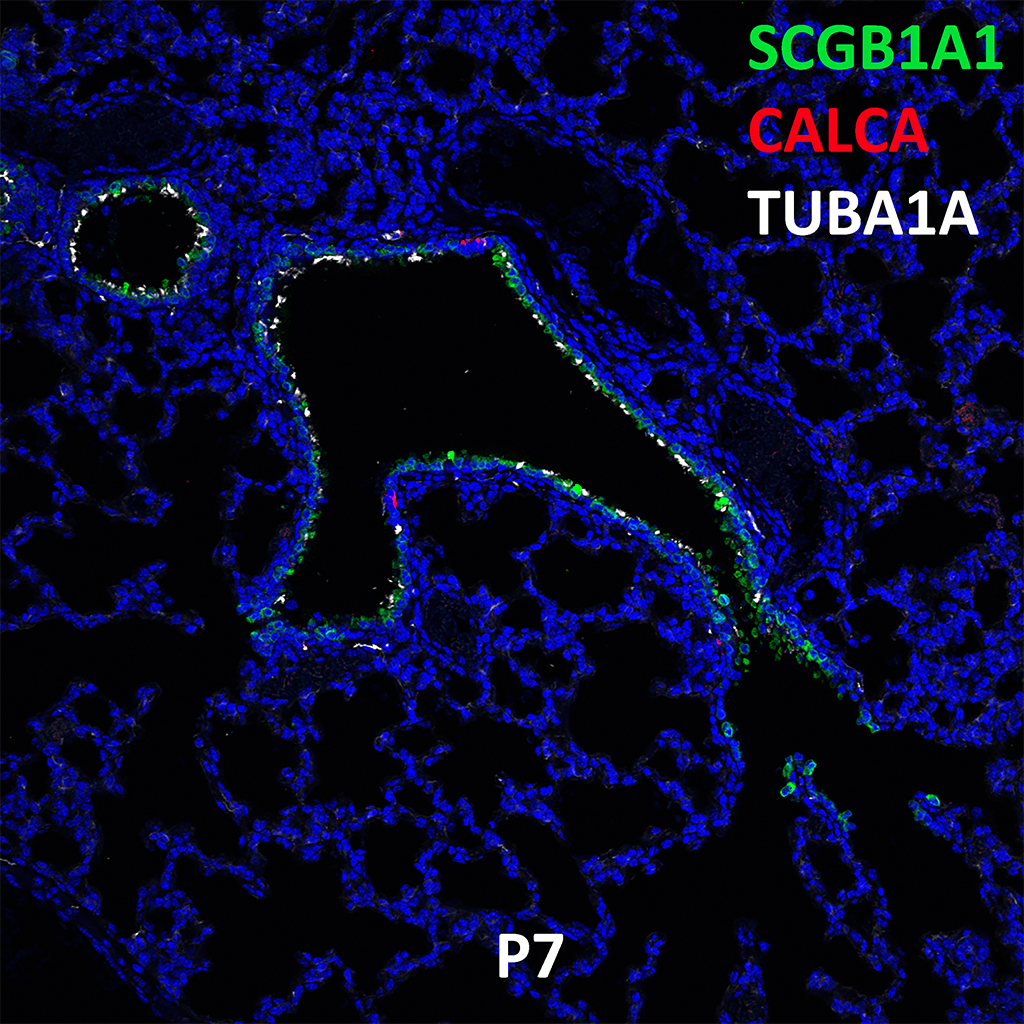
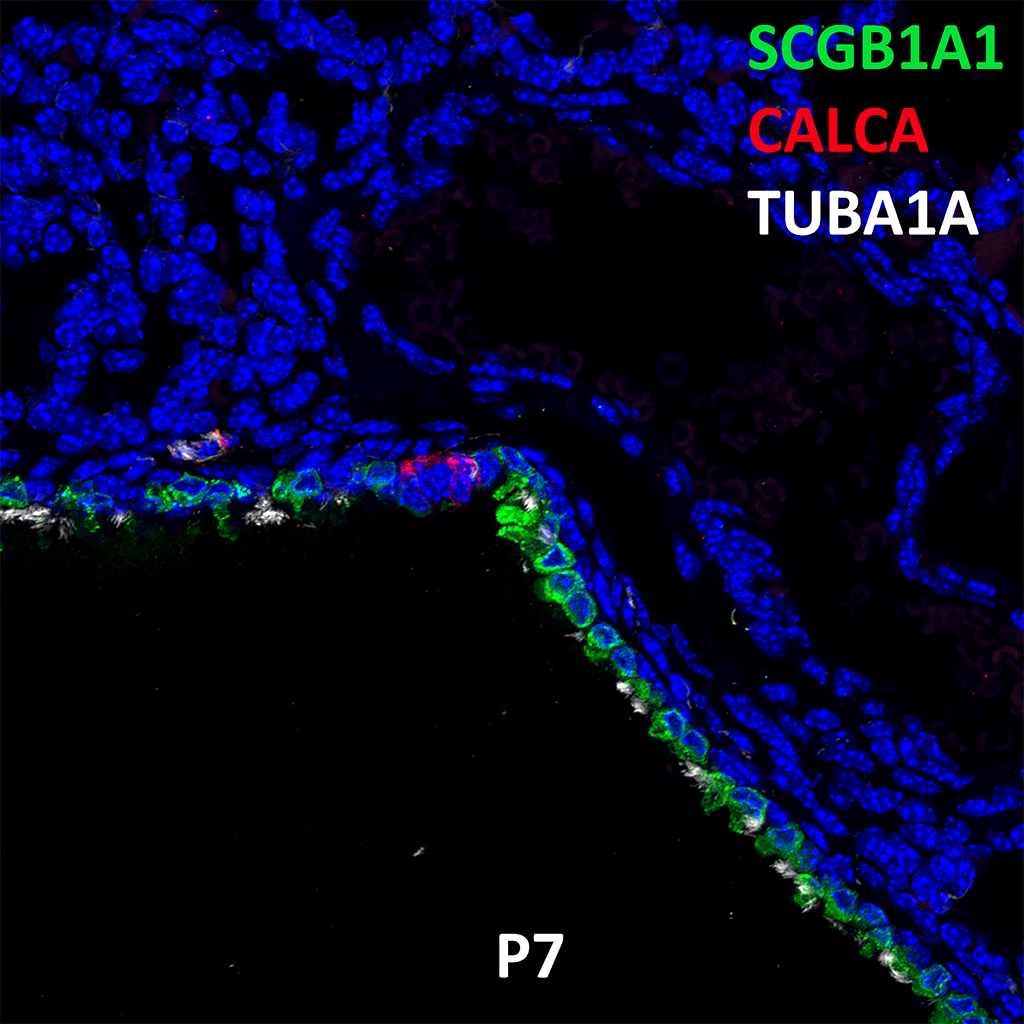
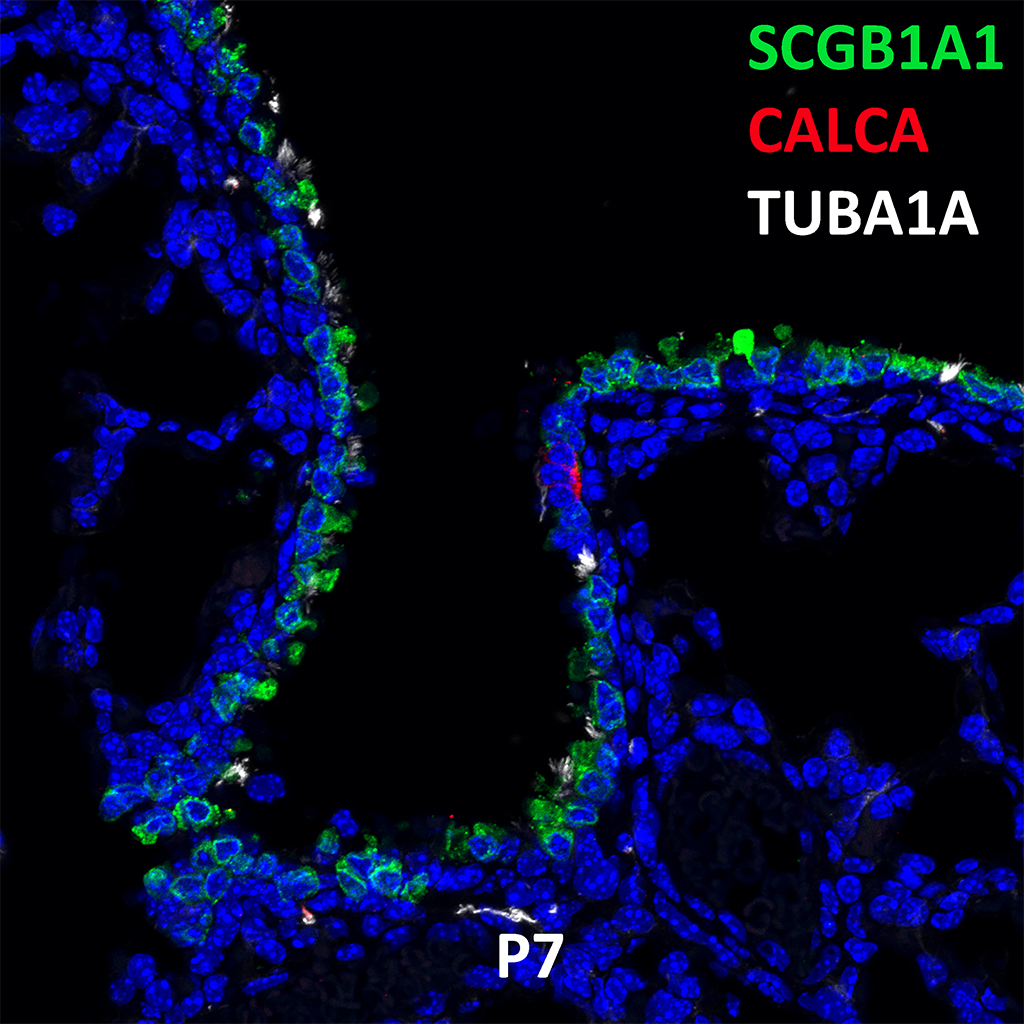
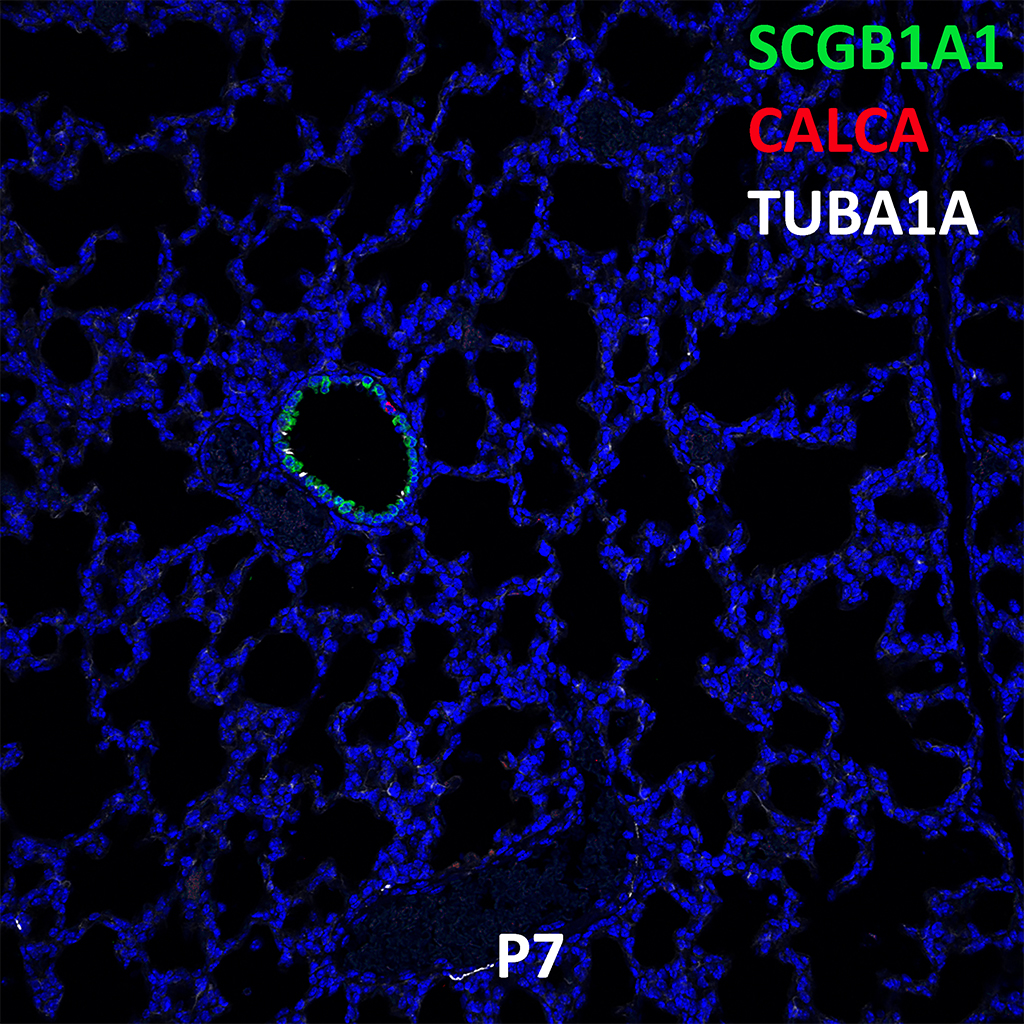
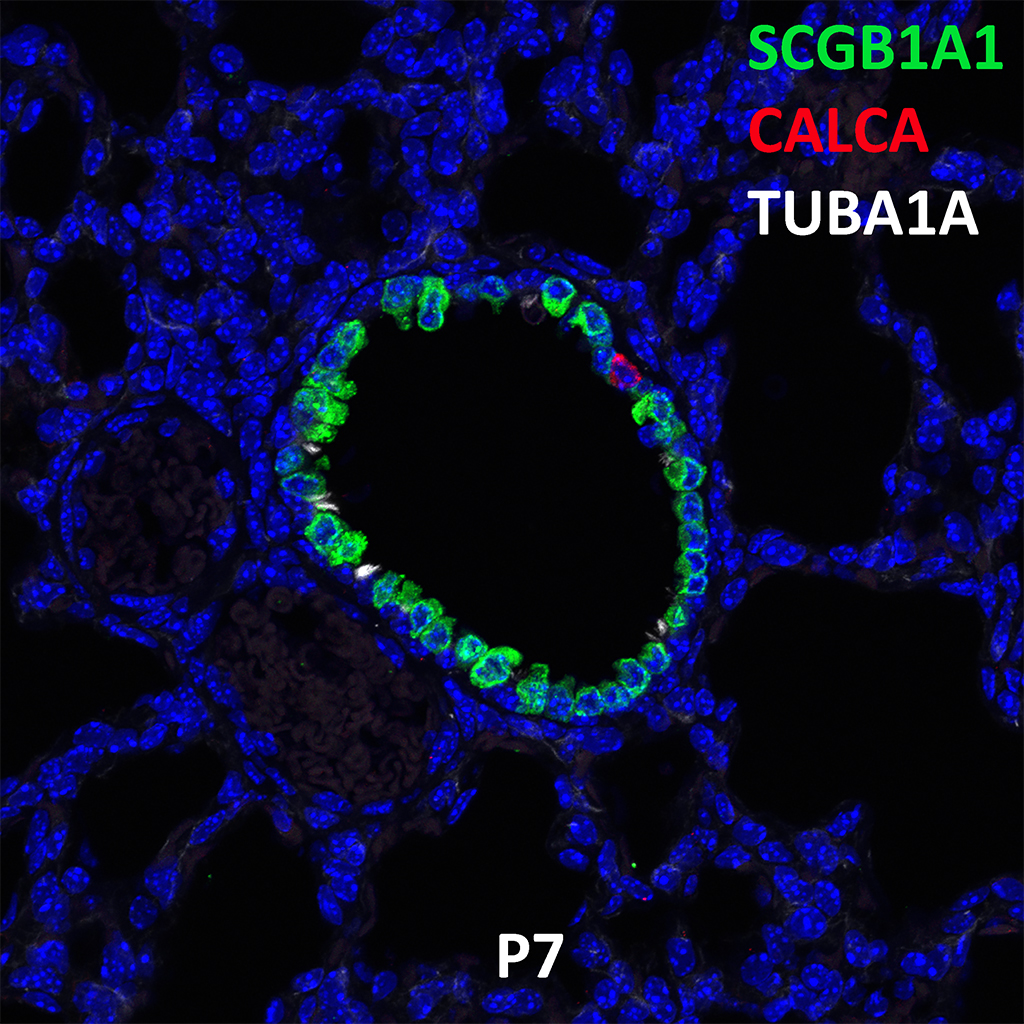
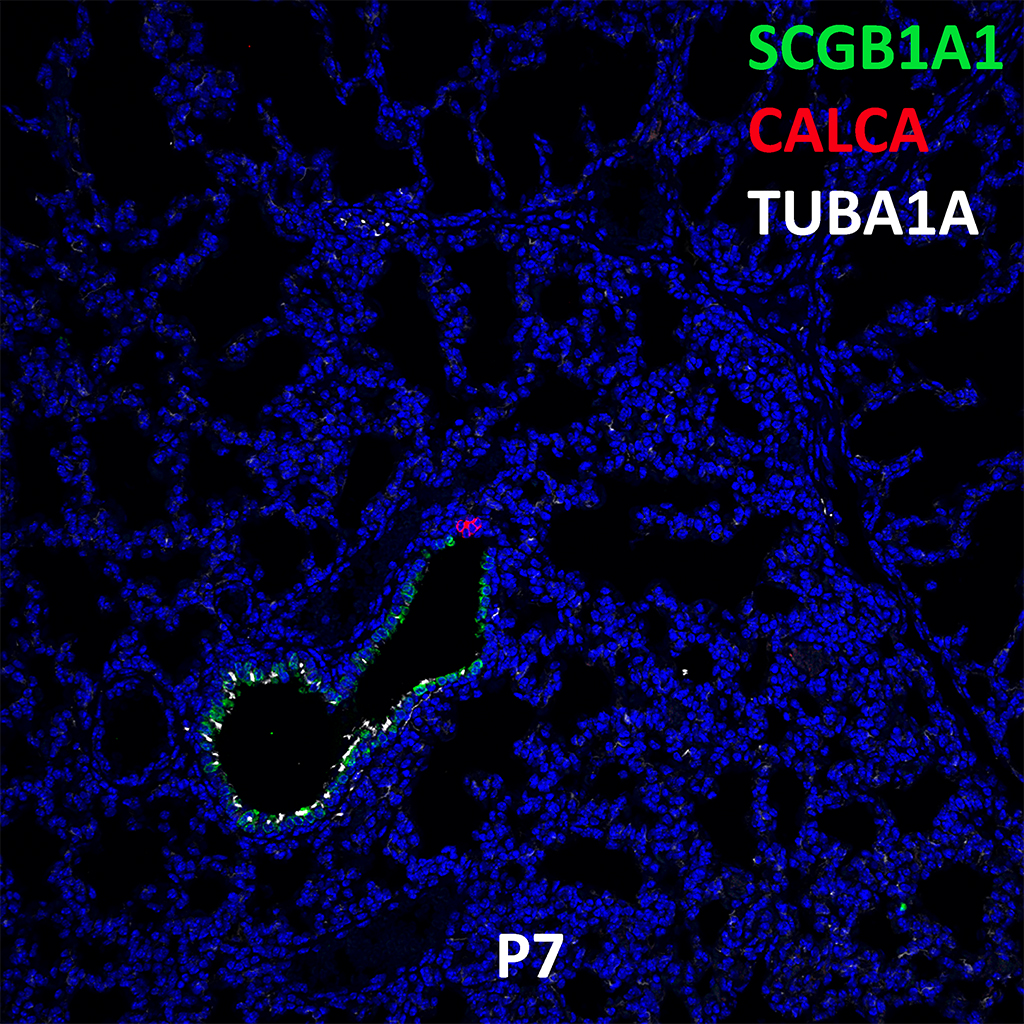
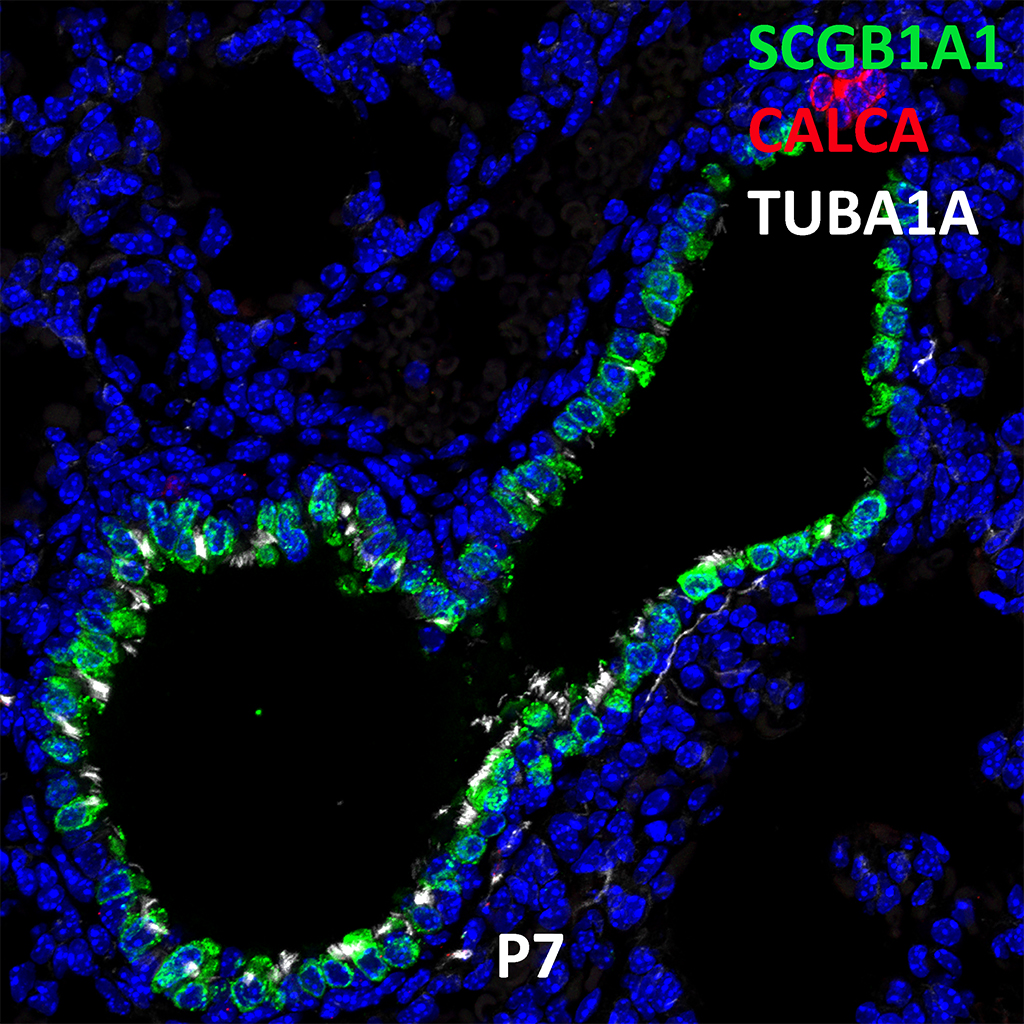
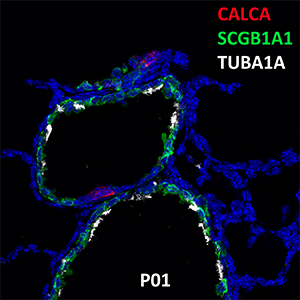
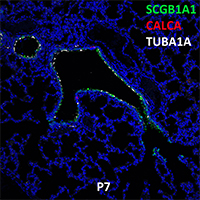
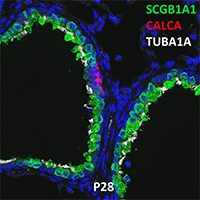
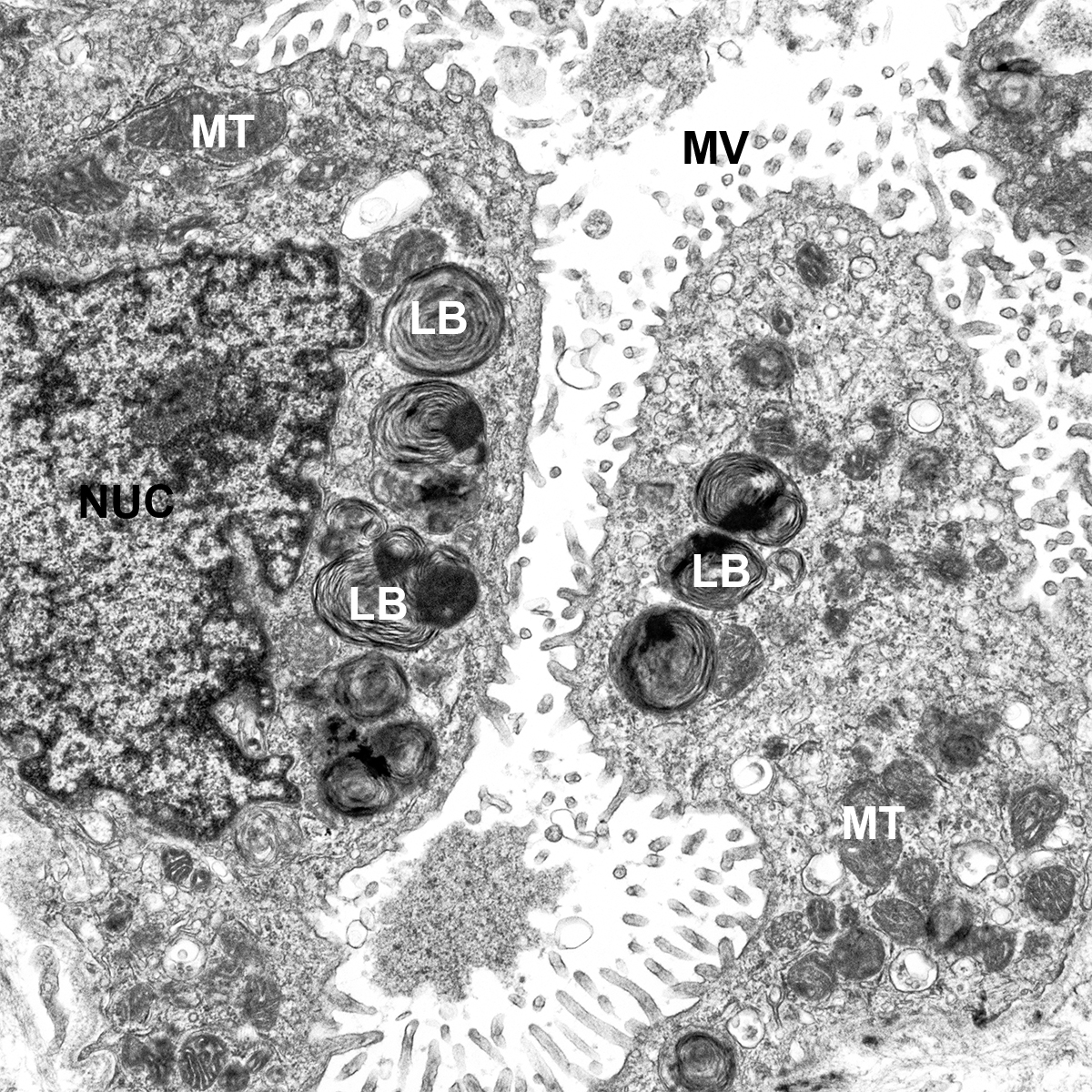
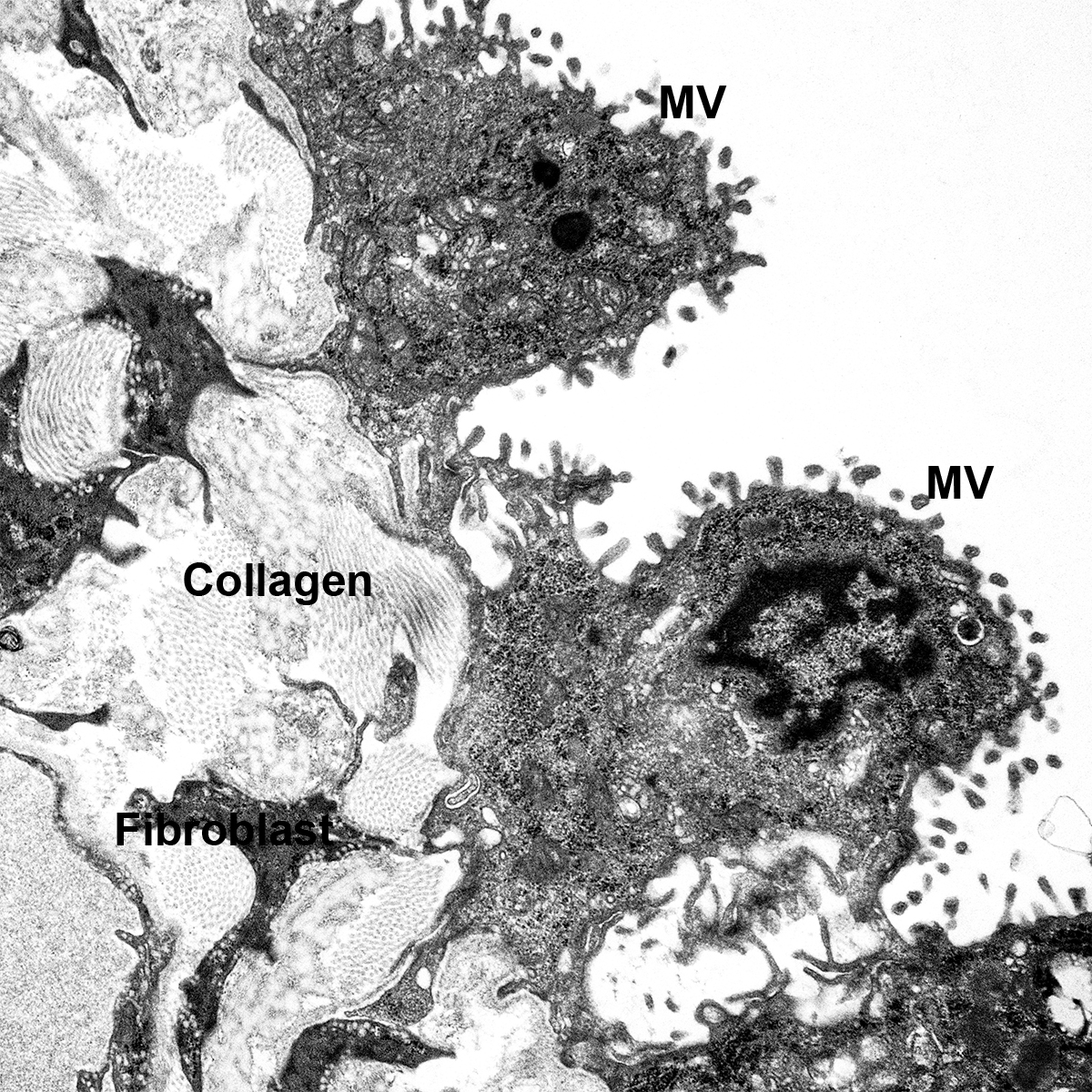
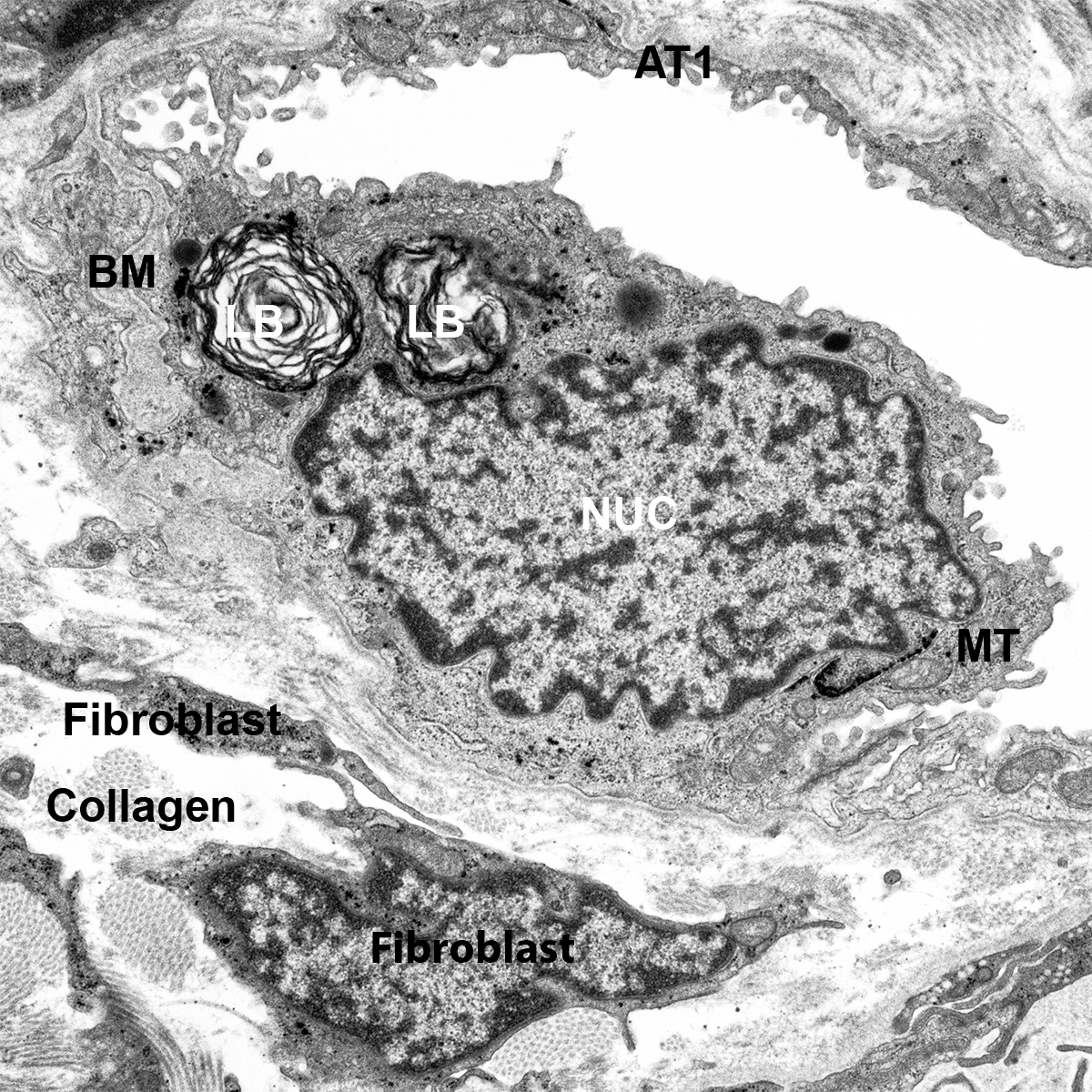
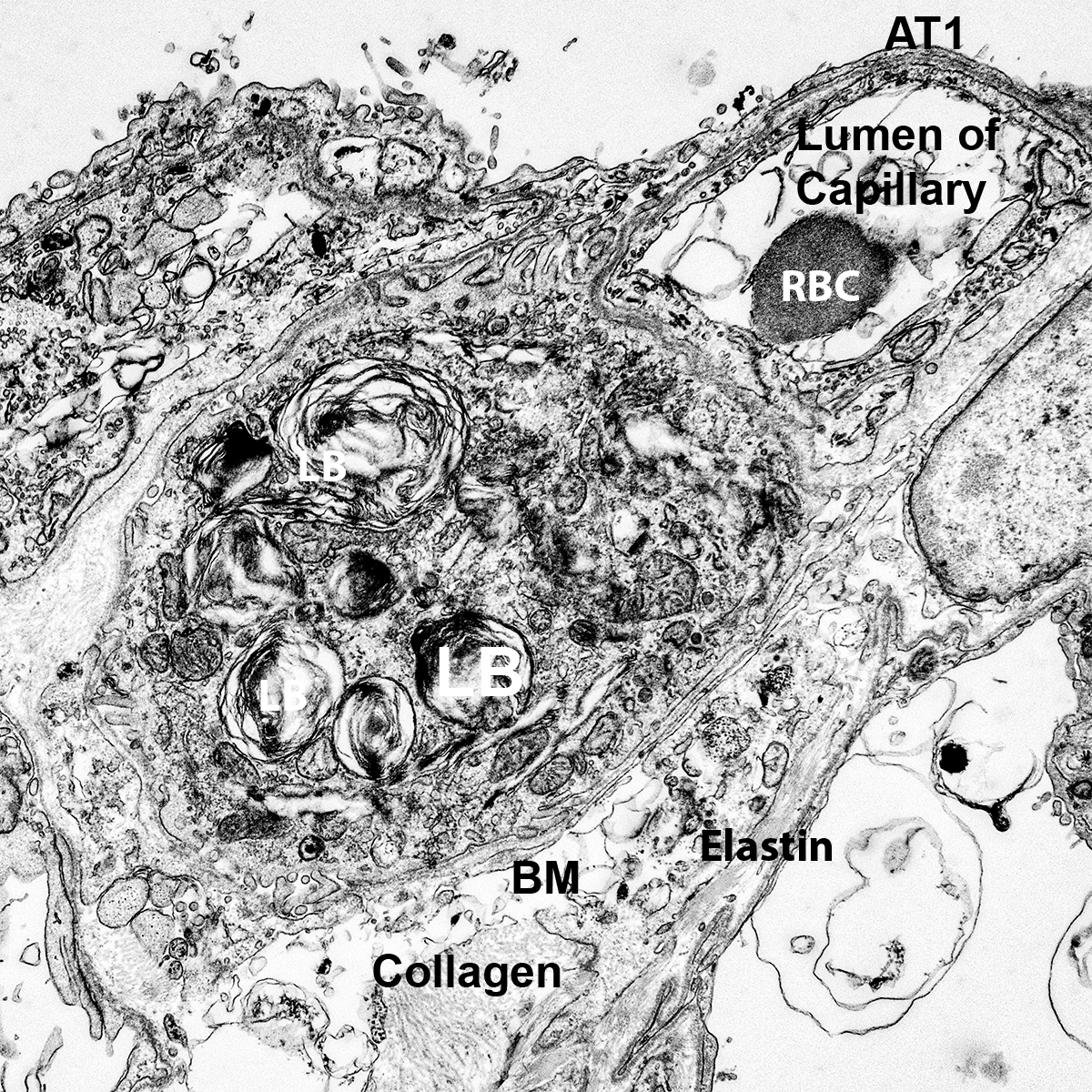
Postnatal Day 28 C57BL6 SCGB1A1, CALCA, and TUBA1A Confocal Imaging
| Gene |
|||||
| Calca | LungGens | Ensemble | Genecards | NCBI | The Human Protein Atlas |
| Scgb1a1a | LungGens | Ensemble |
Genecards | NCBI | The Human Protein Atlas |
| Tuba1a | LungGens | Ensemble | Genecards | NCBI | The Human Protein Atlas |
Day 1
- Paraffin sections are placed at 60oC for 2 hours to overnight to melt paraffin. Paraffin sections are then placed in xylene 3X for 10 minutes each, followed by 3X in 100% ETOH for 3 minutes each, 95% ETOH for 3 minutes each, and 70% ETOH for 3 minutes each. Slides are then placed in 1X PBS for 5 minutes to completely rehydrate tissue.
- Briefly equilibrate slides in Antigen Retrieval Buffer.
- 10 mM sodium citrate, pH 6.0, and heat in a microwave for 15 min at 96oC. *We usually do a series of three runs to equal the time/temp because of evaporation (refill coplin jars with dH2O).
- Microwave according to instructions on microwave.
- Cool on countertop, 15 min.
- Rinse with dH2O.
- 1X PBS, 5 min.
- Block in 4% Goat serum/PBS-T, 2 hours at RT.
- For mouse anti –CALCA (C7113, Sigma) dilute 1:100 in blocking buffer, For guinea pig anti-CCSP (G210, Bl. 3/24/11) dilute 1:1500, for mouse anti- TUBA1A1(T7451, Sigma) dilute 1:1000 in blocking buffer. Spin for 10 minutes in a µfuge, apply to tissue, and incubate on tissue overnight @ 4oC.
Day 2
- Rinse slides in PBS-T 3X, 5 min.
- Apply secondary antibody , Goat Alexa Fluor 488 anti-mouse IgG1 (A21121, Lot# 1345050, for anti-CALCA ) dilute 1:200, Goat Alexa Fluor 555 anti-guinea pig IgG (A21435, Lot# 1571699, CCSP) at 1:200, Donkey Alexa Fluor 647 anti-mouse IgG2B (A21242, Lot# 1148959, anti-TUBA1A1) at 1:200, in blocking buffer. Vortex and µfuge at full speed for 10 minutes. Incubated at room temperature for 1 hour.
- Rinse in PBS-T 3X, 5 min.
- Dilute DAPI 1:2000 and apply to slides for 10 min.
- Wash in PBS-T 3X, 5 min.
- Rinse slides in 0.1M PB or TB, 3X, 5 min.
- Add 1 drop of Prolong Gold anti-fade mounting medium (P36930).
- Coverslip with Gold Seal Coverslip (Cat# 3422 Electron Microscopy Sciences, 22 X 22 mm).
- Allow Prolong Gold to cure overnight at room temp in light sealed box.
Tissue Used:
LMM.15.16A.6.36
PND28
Gender: Female
Weight: 10.0g
Appendix
Antigen Retrieval Solution
18ml Solution A- 0.1M Citric Acid (Sigma, C1909) pH 2.5
82ml Reagent B- 0.1M Sodium Citrate (Sigma, S4641 ) pH 8.2
1L dH2O
pH 6.0